Suppression of Endothelial AGO1 Promotes Adipose Tissue Browning and Improves Metabolic Dysfunction
- PMID: 32393053
- PMCID: PMC7390348
- DOI: 10.1161/CIRCULATIONAHA.119.041231
Suppression of Endothelial AGO1 Promotes Adipose Tissue Browning and Improves Metabolic Dysfunction
Erratum in
-
Correction to: Suppression of Endothelial AGO1 Promotes Adipose Tissue Browning and Improves Metabolic Dysfunction.Circulation. 2021 Apr 20;143(16):e871. doi: 10.1161/CIR.0000000000000977. Epub 2021 Apr 19. Circulation. 2021. PMID: 33872082 No abstract available.
Abstract
Background: Metabolic disorders such as obesity and diabetes mellitus can cause dysfunction of endothelial cells (ECs) and vascular rarefaction in adipose tissues. However, the modulatory role of ECs in adipose tissue function is not fully understood. Other than vascular endothelial growth factor-vascular endothelial growth factor receptor-mediated angiogenic signaling, little is known about the EC-derived signals in adipose tissue regulation. We previously identified Argonaute 1 (AGO1; a key component of microRNA-induced silencing complex) as a crucial regulator in hypoxia-induced angiogenesis. In this study, we intend to determine the AGO1-mediated EC transcriptome, the functional importance of AGO1-regulated endothelial function in vivo, and the relevance to adipose tissue function and obesity.
Methods: We generated and subjected mice with EC-AGO1 deletion (EC-AGO1-knockout [KO]) and their wild-type littermates to a fast food-mimicking, high-fat high-sucrose diet and profiled the metabolic phenotypes. We used crosslinking immunoprecipitation- and RNA-sequencing to identify the AGO1-mediated mechanisms underlying the observed metabolic phenotype of EC-AGO1-KO. We further leveraged cell cultures and mouse models to validate the functional importance of the identified molecular pathway, for which the translational relevance was explored using human endothelium isolated from healthy donors and donors with obesity/type 2 diabetes mellitus.
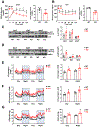
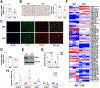
Results: We identified an antiobesity phenotype of EC-AGO1-KO, evident by lower body weight and body fat, improved insulin sensitivity, and enhanced energy expenditure. At the organ level, we observed the most significant phenotype in the subcutaneous and brown adipose tissues of KO mice, with greater vascularity and enhanced browning and thermogenesis. Mechanistically, EC-AGO1 suppression results in inhibition of thrombospondin-1 (THBS1/TSP1), an antiangiogenic and proinflammatory cytokine that promotes insulin resistance. In EC-AGO1-KO mice, overexpression of TSP1 substantially attenuated the beneficial phenotype. In human endothelium isolated from donors with obesity or type 2 diabetes mellitus, AGO1 and THBS1 are expressed at higher levels than the healthy controls, supporting a pathological role of this pathway.
Conclusions: Our study suggests a novel mechanism by which ECs, through the AGO1-TSP1 pathway, control vascularization and function of adipose tissues, insulin sensitivity, and whole-body metabolic state.
Keywords: AGO1; TSP1/THBS1; adipose tissue; angiogenesis; endothelial cells; insulin resistance; obesity.
Figures




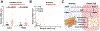
Comment in
-
Endotheliopathy of Obesity.Circulation. 2020 Jul 28;142(4):380-383. doi: 10.1161/CIRCULATIONAHA.120.047574. Epub 2020 Jul 27. Circulation. 2020. PMID: 32718250 Free PMC article. No abstract available.
References
-
- Kahn SE, Hull RL and Utzschneider KM. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature. 2006;444:840–846. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

