Muscovite nanoparticles mitigate neuropathic pain by modulating the inflammatory response and neuroglial activation in the spinal cord
- PMID: 32394976
- PMCID: PMC7716045
- DOI: 10.4103/1673-5374.282260
Muscovite nanoparticles mitigate neuropathic pain by modulating the inflammatory response and neuroglial activation in the spinal cord
Abstract
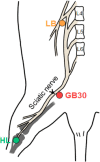
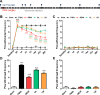
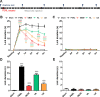
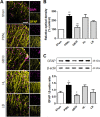
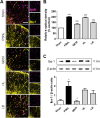
Despite numerous efforts to overcome neuropathic pain, various pharmacological drugs often fail to meet the needs and have many side effects. Muscovite is an aluminosilicate mineral that has been reported to have an anti-inflammatory effect, but the efficacy of muscovite for neuropathic pain has not been investigated. Here, we assessed whether muscovite nanoparticles can reduce the symptoms of pain by controlling the inflammatory process observed in neuropathic pain. The analgesic effects of muscovite nanoparticles were explored using partial sciatic nerve ligation model of neuropathic pain, in which one-third to one-half of the nerve trifurcation of the sciatic nerve was tightly tied to the dorsal side. Muscovite nanoparticles (4 mg/100 μL) was given intramuscularly to evaluate its effects on neuropathic pain (3 days per week for 4 weeks). The results showed that the muscovite nanoparticle injections significantly alleviated partial sciatic nerve ligation-induced mechanical and cold allodynia. In the spinal cord, the muscovite nanoparticle injections exhibited inhibitory effects on astrocyte and microglia activation and reduced the expression of pro-inflammatory cytokines, such as interleukin-1β, tumor necrosis factor-α, interleiukin-6 and monocyte chemoattractant protein-1, which were upregulated in the partial sciatic nerve ligation model. Moreover, the muscovite nanoparticle injections resulted in a decrease in activating transcription factor 3, a neuronal injury marker, in the sciatic nerve. These results suggest that the analgesic effects of muscovite nanoparticle on partial sciatic nerve ligation-induced neuropathic pain may result from inhibiting activation of astrocytes and microglia as well as pro-inflammatory cytokines. We propose that muscovite nanoparticle is a potential anti-nociceptive candidate for neuropathic pain. All experimental protocols in this study were approved by the Institutional Animal Ethics Committee (IACUC) at Dongguk University, South Korea (approval No. 2017-022-1) on September 28, 2017.
Keywords: astrocyte; microglia; muscovite; nanoparticle; neuropathic pain; partial sciatic nerve ligation; pharmacopuncture; pro-inflammatory cytokine; spinal cord.
Conflict of interest statement
None
Figures
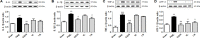
References
-
- Andreev N, Urban L, Dray A. Opioids suppress spontaneous activity of polymodal nociceptors in rat paw skin induced by ultraviolet irradiation. Neuroscience. 1994;58:793–798. - PubMed
-
- Averill S, Michael GJ, Shortland PJ, Leavesley RC, King VR, Bradbury EJ, McMahon SB, Priestley JV. NGF and GDNF ameliorate the increase in ATF3 expression which occurs in dorsal root ganglion cells in response to peripheral nerve injury. Eur J Neurosci. 2004;19:1437–1445. - PubMed
-
- Benyamin R, Trescot AM, Datta S, Buenaventura R, Adlaka R, Sehgal N, Glaser SE, Vallejo R. Opioid complications and side effects. Pain Physician. 2008;11(2 Suppl):S105–120. - PubMed
-
- Calvo M, Dawes JM, Bennett DL. The role of the immune system in the generation of neuropathic pain. Lancet Neurol. 2012;11:629–642. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

