Is the Way to Fight Cancer Paved with Gold? Metal-Based Carbene Complexes with Multiple and Fascinating Biological Features
- PMID: 32403274
- PMCID: PMC7281280
- DOI: 10.3390/ph13050091
Is the Way to Fight Cancer Paved with Gold? Metal-Based Carbene Complexes with Multiple and Fascinating Biological Features
Abstract
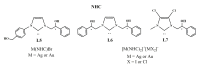
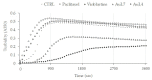
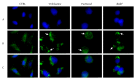
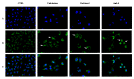
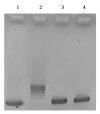
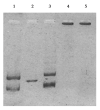
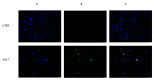
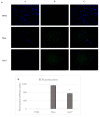
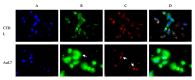
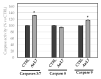
Herein, we report the synthesis and the multiple anti-tumor properties of new gold and silver carbene complexes. The chemical modifications, grounded on our previous studies, led us to identify a good lead complex, gold-based, whose biological features are very exciting and promising in the anti-cancer research and could be further developed. Indeed, the bis-[4,5-dichloro-(N-methyl-N'(2-hydroxy-2-phenyl)ethyl-imidazole-2-ylidene)gold(I)]+[dichloro-gold]- (AuL7) complex possesses the ability to interfere with at least three important and different intracellular targets, namely the human topoisomerases I and II and tubulin, which are able to modulate metabolic processes not directly correlated each other. We proved that the modifications of the ligands structure in AuL7, with respect to another already published complex, i.e., bis-[4,5-dichloro-(N-methyl-N'(cyclopentane-2ol)-imidazole-2-ylidine)gold(I)]+[dichloro-gold]- (AuL4), produce a different behavior toward tubulin-polymerization process, since AuL7 is a tubulin-polymerization inhibitor and AuL4 a stabilizer, with the final same result of hampering the tumor growth. Taken together, our outcomes designate AuL7 as a promising compound for the development of multi-targeted anti-cancer therapies.
Keywords: carbene complexes; docking studies; human topoisomerases I and II; tubulin; women cancers.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
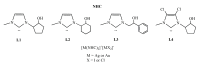

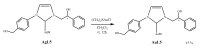
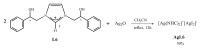
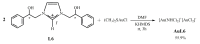


References
-
- Ceramella J., Caruso A., Occhiuzzi M.A., Iacopetta D., Barbarossa A., Rizzuti B., Dallemagne P., Rault S., El-Kashef H., Saturnino C., et al. Benzothienoquinazolinones as new multi-target scaffolds: Dual inhibition of human Topoisomerase I and tubulin polymerization. Eur. J. Med. Chem. 2019;181:111583. doi: 10.1016/j.ejmech.2019.111583. - DOI - PubMed
-
- Aune D., Navarro Rosenblatt D.A., Chan D.S., Abar L., Vingeliene S., Vieira A.R., Greenwood D.C., Norat T. Anthropometric factors and ovarian cancer risk: A systematic review and nonlinear dose-response meta-analysis of prospective studies. Int. J. Cancer. 2015;136:1888–1898. doi: 10.1002/ijc.29207. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

