Detection of SARS-CoV-2-Specific Humoral and Cellular Immunity in COVID-19 Convalescent Individuals
- PMID: 32413330
- PMCID: PMC7196424
- DOI: 10.1016/j.immuni.2020.04.023
Detection of SARS-CoV-2-Specific Humoral and Cellular Immunity in COVID-19 Convalescent Individuals
Abstract
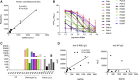
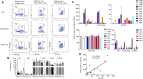
The World Health Organization has declared SARS-CoV-2 virus outbreak a worldwide pandemic. However, there is very limited understanding on the immune responses, especially adaptive immune responses to SARS-CoV-2 infection. Here, we collected blood from COVID-19 patients who have recently become virus-free, and therefore were discharged, and detected SARS-CoV-2-specific humoral and cellular immunity in eight newly discharged patients. Follow-up analysis on another cohort of six patients 2 weeks post discharge also revealed high titers of immunoglobulin G (IgG) antibodies. In all 14 patients tested, 13 displayed serum-neutralizing activities in a pseudotype entry assay. Notably, there was a strong correlation between neutralization antibody titers and the numbers of virus-specific T cells. Our work provides a basis for further analysis of protective immunity to SARS-CoV-2, and understanding the pathogenesis of COVID-19, especially in the severe cases. It also has implications in developing an effective vaccine to SARS-CoV-2 infection.
Keywords: COVID-19 patients; SARS-CoV-2; SARS-CoV-2-specific T cells; SARS-CoV-2-specific antibody; adaptive immunity.
Copyright © 2020 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Interests L.N., Y.F., W.P., and C.D. have filed a provisional patent on the methodology of detecting SARS-CoV-2-specific antibody responses.
Figures

References
-
- Deng H.K., Unutmaz D., KewalRamani V.N., Littman D.R. Expression cloning of new receptors used by simian and human immunodeficiency viruses. Nature. 1997;388:296–300. - PubMed
-
- Di Pierro F., Bertuccioli A., Cavecchia I. Possible therapeutic role of a highly standardized mixture of active compounds derived from cultured Lentinula edodes mycelia (AHCC) in patients infected with 2019 novel coronavirus. Minerva Gastroenterol. Dietol. 2020 doi: 10.23736/S1121-421X.20.02697-5. Published online March 12, 2020. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

