Functional stability of water wire-carbonyl interactions in an ion channel
- PMID: 32414918
- PMCID: PMC7275723
- DOI: 10.1073/pnas.2001083117
Functional stability of water wire-carbonyl interactions in an ion channel
Abstract
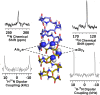
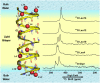
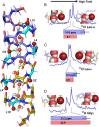
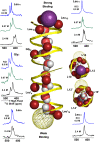
Water wires are critical for the functioning of many membrane proteins, as in channels that conduct water, protons, and other ions. Here, in liquid crystalline lipid bilayers under symmetric environmental conditions, the selective hydrogen bonding interactions between eight waters comprising a water wire and a subset of 26 carbonyl oxygens lining the antiparallel dimeric gramicidin A channel are characterized by 17O NMR spectroscopy at 35.2 T (or 1,500 MHz for 1H) and computational studies. While backbone 15N spectra clearly indicate structural symmetry between the two subunits, single site 17O labels of the pore-lining carbonyls report two resonances, implying a break in dimer symmetry caused by the selective interactions with the water wire. The 17O shifts document selective water hydrogen bonding with carbonyl oxygens that are stable on the millisecond timescale. Such interactions are supported by density functional theory calculations on snapshots taken from molecular dynamics simulations. Water hydrogen bonding in the pore is restricted to just three simultaneous interactions, unlike bulk water environs. The stability of the water wire orientation and its electric dipole leads to opposite charge-dipole interactions for K+ ions bound at the two ends of the pore, thereby providing a simple explanation for an ∼20-fold difference in K+ affinity between two binding sites that are ∼24 Å apart. The 17O NMR spectroscopy reported here represents a breakthrough in high field NMR technology that will have applications throughout molecular biophysics, because of the acute sensitivity of the 17O nucleus to its chemical environment.
Keywords: 17O NMR; gramicidin A; molecular dynamics; ultra-high field NMR; water wire.
Copyright © 2020 the Author(s). Published by PNAS.
Conflict of interest statement
The authors declare no competing interest.
Figures
Similar articles
-
Structure and dynamics of a proton wire: a theoretical study of H+ translocation along the single-file water chain in the gramicidin A channel.Biophys J. 1996 Jul;71(1):19-39. doi: 10.1016/S0006-3495(96)79211-1. Biophys J. 1996. PMID: 8804586 Free PMC article.
-
Ion solvation by channel carbonyls characterized by 17O solid-state NMR at 21 T.J Am Chem Soc. 2005 Aug 31;127(34):11922-3. doi: 10.1021/ja0535413. J Am Chem Soc. 2005. PMID: 16117514
-
Molecular mechanism of H+ conduction in the single-file water chain of the gramicidin channel.Biophys J. 2002 May;82(5):2304-16. doi: 10.1016/S0006-3495(02)75576-8. Biophys J. 2002. PMID: 11964221 Free PMC article.
-
Correlations of structure, dynamics and function in the gramicidin channel by solid-state NMR spectroscopy.Novartis Found Symp. 1999;225:4-16; discussion 16-22. Novartis Found Symp. 1999. PMID: 10472044 Review.
-
Gramicidin channels.IEEE Trans Nanobioscience. 2005 Mar;4(1):10-20. doi: 10.1109/tnb.2004.842470. IEEE Trans Nanobioscience. 2005. PMID: 15816168 Review.
Cited by
-
Surprising Rigidity of Functionally Important Water Molecules Buried in the Lipid Headgroup Region.J Am Chem Soc. 2022 May 4;144(17):7881-7888. doi: 10.1021/jacs.2c02145. Epub 2022 Apr 19. J Am Chem Soc. 2022. PMID: 35439409 Free PMC article.
-
Field-stepped ultra-wideline NMR at up to 36 T: On the inequivalence between field and frequency stepping.Magn Reson Chem. 2021 Sep;59(9-10):951-960. doi: 10.1002/mrc.5128. Epub 2021 Jan 12. Magn Reson Chem. 2021. PMID: 33373086 Free PMC article.
-
1H/17O Chemical Shift Waves in Carboxyl-Bridged Hydrogen Bond Networks in Organic Solids.J Phys Chem A. 2024 May 30;128(21):4288-4296. doi: 10.1021/acs.jpca.4c01866. Epub 2024 May 15. J Phys Chem A. 2024. PMID: 38748612 Free PMC article.
-
Polarization Effects in Water-Mediated Selective Cation Transport across a Narrow Transmembrane Channel.J Chem Theory Comput. 2021 Mar 9;17(3):1726-1741. doi: 10.1021/acs.jctc.0c00968. Epub 2021 Feb 4. J Chem Theory Comput. 2021. PMID: 33539082 Free PMC article.
-
Characterization of peptide O⋯HN hydrogen bonds via1H-detected 15N/17O solid-state NMR spectroscopy.Chem Commun (Camb). 2023 Mar 9;59(21):3111-3113. doi: 10.1039/d2cc07004a. Chem Commun (Camb). 2023. PMID: 36804656 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

