Patterns of Cortical Folding Associated with Autistic Symptoms in Carriers and Noncarriers of the 22q11.2 Microdeletion
- PMID: 32420595
- PMCID: PMC7566689
- DOI: 10.1093/cercor/bhaa108
Patterns of Cortical Folding Associated with Autistic Symptoms in Carriers and Noncarriers of the 22q11.2 Microdeletion
Erratum in
-
Corrigendum to: Patterns of Cortical Folding Associated with Autistic Symptoms in Carriers and Noncarriers of the 22q11.2 Microdeletion.Cereb Cortex. 2020 Jul 30;30(9):5191. doi: 10.1093/cercor/bhaa194. Cereb Cortex. 2020. PMID: 32609321 Free PMC article. No abstract available.
Abstract
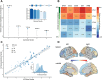
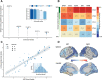
22q11.2 deletion syndrome (22q11.2DS) is a genetic condition accompanied by a range of psychiatric manifestations, including autism spectrum disorder (ASD). It remains unknown, however, whether these symptoms are mediated by the same or distinct neural mechanisms as in idiopathic ASD. Here, we examined differences in lGI associated with ASD in 50 individuals with 22q11.2DS (n = 25 with ASD, n = 25 without ASD) and 81 individuals without 22q11.2DS (n = 40 with ASD, n = 41 typically developing controls). We initially utilized a factorial design to identify the set of brain regions where lGI is associated with the main effect of 22q11.2DS, ASD, and with the 22q11.2DS-by-ASD interaction term. Subsequently, we employed canonical correlation analysis (CCA) to compare the multivariate association between variability in lGI and the complex clinical phenotype of ASD between 22q11.2DS carriers and noncarriers. Across approaches, we established that even though there is a high degree of clinical similarity across groups, the associated patterns of lGI significantly differed between carriers and noncarriers of the 22q11.2 microdeletion. Our results suggest that ASD symptomatology recruits different neuroanatomical underpinnings across disorders and that 22q11.2DS individuals with ASD represent a neuroanatomically distinct subgroup that differs from 22q11.2DS individuals without ASD and from individuals with idiopathic ASD.
Keywords: 22q11.2 deletion syndrome; autism spectrum disorder; brain anatomy; cortical folding; local gyrification index.
© The Author(s) 2020. Published by Oxford University Press. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures


Similar articles
-
Neuroanatomical underpinnings of autism symptomatology in carriers and non-carriers of the 22q11.2 microdeletion.Mol Autism. 2020 Jun 8;11(1):46. doi: 10.1186/s13229-020-00356-z. Mol Autism. 2020. PMID: 32513259 Free PMC article.
-
The Neuroanatomy of Autism Spectrum Disorder Symptomatology in 22q11.2 Deletion Syndrome.Cereb Cortex. 2019 Jul 22;29(8):3655-3665. doi: 10.1093/cercor/bhy239. Cereb Cortex. 2019. PMID: 30272146 Free PMC article.
-
22q11.2 duplication syndrome: elevated rate of autism spectrum disorder and need for medical screening.Mol Autism. 2016 May 6;7:27. doi: 10.1186/s13229-016-0090-z. eCollection 2016. Mol Autism. 2016. PMID: 27158440 Free PMC article.
-
[Neurocognitive and psychiatric management of the 22q11.2 deletion syndrome].Encephale. 2015 Jun;41(3):266-73. doi: 10.1016/j.encep.2014.10.005. Epub 2014 Dec 16. Encephale. 2015. PMID: 25523123 Review. French.
-
Developmental trajectories in 22q11.2 deletion.Am J Med Genet C Semin Med Genet. 2015 Jun;169(2):172-81. doi: 10.1002/ajmg.c.31435. Epub 2015 May 18. Am J Med Genet C Semin Med Genet. 2015. PMID: 25989227 Free PMC article. Review.
Cited by
-
Neuroimaging Phenotypes Associated With Risk and Resilience for Psychosis and Autism Spectrum Disorders in 22q11.2 Microdeletion Syndrome.Biol Psychiatry Cogn Neurosci Neuroimaging. 2021 Feb;6(2):211-224. doi: 10.1016/j.bpsc.2020.08.015. Epub 2020 Sep 6. Biol Psychiatry Cogn Neurosci Neuroimaging. 2021. PMID: 33218931 Free PMC article. Review.
-
An exploratory fetal MRI study examining the impact of 22q11.2 microdeletion syndrome on early brain growth.J Neurodev Disord. 2025 Feb 12;17(1):7. doi: 10.1186/s11689-025-09594-9. J Neurodev Disord. 2025. PMID: 39939911 Free PMC article.
-
Neuroanatomical Correlates of Cognitive Dysfunction in 22q11.2 Deletion Syndrome.Genes (Basel). 2024 Mar 30;15(4):440. doi: 10.3390/genes15040440. Genes (Basel). 2024. PMID: 38674375 Free PMC article. Review.
-
Brain morphometry in 22q11.2 deletion syndrome: an exploration of differences in cortical thickness, surface area, and their contribution to cortical volume.Sci Rep. 2020 Nov 2;10(1):18845. doi: 10.1038/s41598-020-75811-1. Sci Rep. 2020. PMID: 33139857 Free PMC article.
References
-
- Antshel KM, Aneja A, Leslie AE, Strunge L, Peebles J, Fremont WP, Stallone K, Abdulsabur N, Higgins AM, Shprintzen RJ, et al. . 2007. Autistic Spectrum disorders in Velo-cardio facial syndrome (22q11.2 deletion). J Autism Dev Disord. 37:1776–1786. - PubMed
-
- Bakker G, Caan MWA, Vingerhoets WAM, Da Silva-Alves F, De Koning M, Boot E, Nieman DH, De Haan L, Bloemen OJ, Booij J, et al. . 2016. Cortical morphology differences in subjects at increased vulnerability for developing a psychotic disorder: a comparison between subjects with ultra-high risk and 22q11.2 deletion syndrome. PLoS One. 11:1–16. - PMC - PubMed
-
- Bölte S, Poustka F. 2008. Skala zur Erfassung sozialer Reaktivität: dimensionale Autismus-Diagnostik; SRS; deutsche Fassung der Social Responsiveness Scale (SRS) von John N. Gruber, Huber: Constantino und Christan P.
-
- Castelli F, Frith C, Happe F, Frith U. 2002. Autism, Asperger syndrome and brain mechanisms for the attribution of mental states to animated shapes. Brain. 125:1839–1849. - PubMed

