Pembrolizumab as First-line Therapy in Cisplatin-ineligible Advanced Urothelial Cancer (KEYNOTE-052): Outcomes in Older Patients by Age and Performance Status
- PMID: 32423837
- PMCID: PMC8246631
- DOI: 10.1016/j.euo.2020.02.009
Pembrolizumab as First-line Therapy in Cisplatin-ineligible Advanced Urothelial Cancer (KEYNOTE-052): Outcomes in Older Patients by Age and Performance Status
Abstract
Background: Patients with treatment-naive advanced urothelial cancer (UC) ineligible for cisplatin-based chemotherapy are typically older and have comorbidities, representing a difficult-to-treat population.
Objective: To evaluate the safety and antitumor activity of first-line pembrolizumab in subgroups of cisplatin-ineligible older patients (aged ≥65 and ≥75 yr) with advanced UC in KEYNOTE-052 (NCT02335424), including those with poor performance status (Eastern Cooperative Oncology Group performance status score 2 [ECOG PS2]).
Design, setting, and participants: Patients were cisplatin ineligible, had treatment-naive, histologically/cytologically confirmed, locally advanced/metastatic UC with measurable disease (Response Evaluation Criteria in Solid Tumors version 1.1 [RECIST v1.1]), and had ECOG PS0-2. Patient subgroups analyzed were aged ≥65yr (n = 302), ≥75 yr (n = 179), ≥65yr with ECOG PS2 (≥65yr+ECOG PS2; n = 119), and ≥75 yr+ECOG PS2 (n = 78).
Intervention: All patients received pembrolizumab 200mg intravenously every 3 wk until confirmed progression, intolerable toxicity, patient withdrawal, or 24 mo of therapy.
Outcome measurements and statistical analysis: The primary endpoint was objective response rate (ORR) as per RECIST v1.1. The key secondary endpoints were overall survival (OS), duration of response (DOR), and safety.
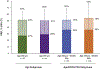
Results and limitations: ORRs for the ≥65yr, ≥75 yr, ≥65yr+ECOG PS2, and ≥75 yr+ECOG PS2 subgroups were 29%, 27%, 29%, and 31%, respectively; rates of complete and partial responses were similar across subgroups (9%, 5%, 6%, and 6%, and 20%, 22%, 23%, and 24%, respectively). Median DOR and OS were also consistent across the ≥65yr and ≥65yr+ECOG PS2 subgroups and the ≥75 yr and ≥75 yr+ECOG PS2 subgroups. Study limitations included open-label design, lack of a comparator group, and nature of post hoc exploratory analysis.
Conclusions: The clinical benefit of pembrolizumab in advanced UC appeared to be consistent regardless of age and/or poor performance status.
Patient summary: This study looked at whether older age and poorer performance status affect how well patients with previously untreated advanced urothelial cancer ineligible for standard-of-care treatment respond to pembrolizumab. Outcomes with pembrolizumab were not affected by older age or poorer performance status, making it an effective option.
Keywords: Aged; Bladder cancer; Checkpoint inhibitor; Cisplatin ineligible; Immunotherapy; Pembrolizumab; Platinum ineligible; Poor performance status; Programmed death 1; Urothelial carcinoma.
Copyright © 2020 The Authors. Published by Elsevier B.V. All rights reserved.
Figures
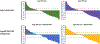
References
-
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018;68:394–424. - PubMed
-
- Galsky MD, Hahn NM, Rosenberg J, et al. Treatment of patients with metastatic urothelial cancer “unfit” for cisplatin-based chemotherapy. J Clin Oncol 2011;29:2432–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

