Comparison Between O and OH Intermediates of Cytochrome c Oxidase Studied by FTIR Spectroscopy
- PMID: 32432087
- PMCID: PMC7215072
- DOI: 10.3389/fchem.2020.00387
Comparison Between O and OH Intermediates of Cytochrome c Oxidase Studied by FTIR Spectroscopy
Abstract
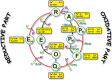
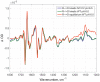
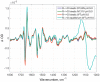
Cytochrome c oxidase is terminal enzyme in the respiratory chain of mitochondria and many aerobic bacteria. It catalyzes reduction of oxygen to water. During its catalysis, CcO proceeds through several quite stable intermediates (R, A, PR/M, O/OH, E/EH). This work is concentrated on the elucidation of the differences between structures of oxidized intermediates O and O H in different CcO variants and at different pH values. Oxidized intermediates of wild type and mutated CcO from Paracoccus denitrificans were studied by means of static and time-resolved Fourier-transform infrared spectroscopy in acidic and alkaline conditions in the infrared region 1800-1000 cm-1. No reasonable differences were found between all variants in these conditions, and in this spectral region. This finding means that the binuclear center of oxygen reduction keeps a very similar structure and holds the same ligands in the studied conditions. The further investigation in search of differences should be performed in the 4000-2000 cm-1 IR region where water ligands absorb.
Keywords: FTIR spectroscopy; cell respiration; cytochrome c oxidase; oxidized state; oxidoreduction.
Copyright © 2020 Gorbikova and Kalendar.
Figures
Similar articles
-
ATR-FTIR spectroscopy of the P(M) and F intermediates of bovine and Paracoccus denitrificans cytochrome c oxidase.Biochemistry. 2003 Jul 29;42(29):8809-17. doi: 10.1021/bi034522d. Biochemistry. 2003. PMID: 12873142
-
Protolytic reactions on reduction of cytochrome c oxidase studied by ATR-FTIR spectroscopy.Biochemistry. 2007 Apr 3;46(13):4177-83. doi: 10.1021/bi602523a. Epub 2007 Mar 7. Biochemistry. 2007. PMID: 17341097
-
ATR-FTIR spectroscopy and isotope labeling of the PM intermediate of Paracoccus denitrificans cytochrome c oxidase.Biochemistry. 2004 Nov 16;43(45):14370-8. doi: 10.1021/bi048545j. Biochemistry. 2004. PMID: 15533041
-
Structures of reaction intermediates of bovine cytochrome c oxidase probed by time-resolved vibrational spectroscopy.J Inorg Biochem. 2000 Nov;82(1-4):9-18. doi: 10.1016/s0162-0134(00)00155-0. J Inorg Biochem. 2000. PMID: 11132644 Review.
-
Respiratory conservation of energy with dioxygen: cytochrome C oxidase.Met Ions Life Sci. 2015;15:89-130. doi: 10.1007/978-3-319-12415-5_4. Met Ions Life Sci. 2015. PMID: 25707467 Review.
Cited by
-
Probing the Proton-Loading Site of Cytochrome C Oxidase Using Time-Resolved Fourier Transform Infrared Spectroscopy.Molecules. 2020 Jul 27;25(15):3393. doi: 10.3390/molecules25153393. Molecules. 2020. PMID: 32727022 Free PMC article.
References
-
- Gobrikova E. (2009). Oxygen Reduction and Proton Translocation by Cytochrome C Oxidase (Ph.D. thesis). University of Helsinki, Department of Biological and Environmental Sciences & Institute of Biotechnology & National Graduate School in Informational and Structural Biology Åbo Akademi, Helsinki, Finland.
LinkOut - more resources
Full Text Sources
Research Materials

