Regulation and dynamics of force transmission at individual cell-matrix adhesion bonds
- PMID: 32440534
- PMCID: PMC7228748
- DOI: 10.1126/sciadv.aax0317
Regulation and dynamics of force transmission at individual cell-matrix adhesion bonds
Abstract
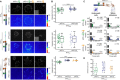
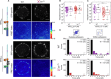
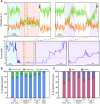
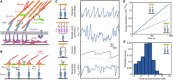
Integrin-based adhesion complexes link the cytoskeleton to the extracellular matrix (ECM) and are central to the construction of multicellular animal tissues. How biological function emerges from the tens to thousands of proteins present within a single adhesion complex remains unclear. We used fluorescent molecular tension sensors to visualize force transmission by individual integrins in living cells. These measurements revealed an underlying functional modularity in which integrin class controlled adhesion size and ECM ligand specificity, while the number and type of connections between integrins and F-actin determined the force per individual integrin. In addition, we found that most integrins existed in a state of near-mechanical equilibrium, a result not predicted by existing models of cytoskeletal force transduction. A revised model that includes reversible cross-links within the F-actin network can account for this result and suggests one means by which cellular mechanical homeostasis can arise at the molecular level.
Copyright © 2020 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works. Distributed under a Creative Commons Attribution NonCommercial License 4.0 (CC BY-NC).
Figures

References
-
- Paszek M. J., Zahir N., Johnson K. R., Lakins J. N., Rozenberg G. I., Gefen A., Reinhart-King C. A., Margulies S. S., Dembo M., Boettiger D., Hammer D. A., Weaver V. M., Tensional homeostasis and the malignant phenotype. Cancer Cell 8, 241–254 (2005). - PubMed
-
- Mekhdjian A. H., Kai F., Rubashkin M. G., Prahl L. S., Przybyla L. M., McGregor A. L., Bell E. S., Barnes J. M., DuFort C. C., Ou G., Chang A. C., Cassereau L., Tan S. J., Pickup M. W., Lakins J. N., Ye X., Davidson M. W., Lammerding J., Odde D. J., Dunn A. R., Weaver V. M., Integrin-mediated traction force enhances paxillin molecular associations and adhesion dynamics that increase the invasiveness of tumor cells into a three-dimensional extracellular matrix. Mol. Biol. Cell 28, 1467–1488 (2017). - PMC - PubMed
-
- Bökel C., Brown N. H., Integrins in development: Moving on, responding to, and sticking to the extracellular matrix. Dev. Cell 3, 311–321 (2002). - PubMed
-
- Horton E. R., Byron A., Askari J. A., Ng D. H. J., Millon-Frémillon A., Robertson J., Koper E. J., Paul N. R., Warwood S., Knight D., Humphries J. D., Humphries M. J., Definition of a consensus integrin adhesome and its dynamics during adhesion complex assembly and disassembly. Nat. Cell Biol. 17, 1577–1587 (2015). - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

