A Selective Serotonin Reuptake Inhibitor, a Proton Pump Inhibitor, and Two Calcium Channel Blockers Inhibit Candida albicans Biofilms
- PMID: 32443498
- PMCID: PMC7285287
- DOI: 10.3390/microorganisms8050756
A Selective Serotonin Reuptake Inhibitor, a Proton Pump Inhibitor, and Two Calcium Channel Blockers Inhibit Candida albicans Biofilms
Abstract
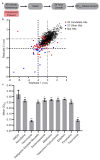
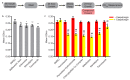
Biofilms formed by the human fungal pathogen Candida albicans are naturally resistant to many of the antifungal agents commonly used in the clinic. We screened a library containing 1600 clinically tested drug compounds to identify compounds that inhibit C. albicans biofilm formation. The compounds that emerged from the initial screen were validated in a secondary screen and then tested for (1) their abilities to disrupt mature biofilms and (2) for synergistic interactions with representatives of the three antifungal agents most commonly prescribed to treat Candida infections, fluconazole, amphotericin B, and caspofungin. Twenty compounds had antibiofilm activity in at least one of the secondary assays and several affected biofilms but, at the same concentration, had little or no effect on planktonic (suspension) growth of C. albicans. Two calcium channel blockers, a selective serotonin reuptake inhibitor, and an azole-based proton pump inhibitor were among the hits, suggesting that members of these three classes of drugs or their derivatives may be useful for treating C. albicans biofilm infections.
Keywords: Candida albicans; Pharmakon 1600 compound library; antimicrobial resistance; biofilm disruption; biofilm inhibition; biofilms; drug repurposing; high-throughput screens; therapeutics.
Conflict of interest statement
Clarissa J. Nobile and Alexander D. Johnson are cofounders of BioSynesis, Inc., a company developing inhibitors and diagnostics of
Figures


References
-
- Kullberg B.J., Oude Lashof A.M.L. Epidemiology of opportunistic invasive mycoses. Eur. J. Med. Res. 2002;7:183–191. - PubMed

