Extra-telomeric impact of telomeres: Emerging molecular connections in pluripotency or stemness
- PMID: 32444498
- PMCID: PMC7383370
- DOI: 10.1074/jbc.REV119.009710
Extra-telomeric impact of telomeres: Emerging molecular connections in pluripotency or stemness
Abstract
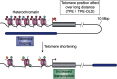
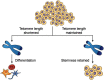
Telomeres comprise specialized nucleic acid-protein complexes that help protect chromosome ends from DNA damage. Moreover, telomeres associate with subtelomeric regions through looping. This results in altered expression of subtelomeric genes. Recent observations further reveal telomere length-dependent gene regulation and epigenetic modifications at sites spread across the genome and distant from telomeres. This regulation is mediated through the telomere-binding protein telomeric repeat-binding factor 2 (TRF2). These observations suggest a role of telomeres in extra-telomeric functions. Most notably, telomeres have a broad impact on pluripotency and differentiation. For example, cardiomyocytes differentiate with higher efficacy from induced pluripotent stem cells having long telomeres, and differentiated cells obtained from human embryonic stem cells with relatively long telomeres have a longer lifespan. Here, we first highlight reports on these two seemingly distinct research areas: the extra-telomeric role of telomere-binding factors and the role of telomeres in pluripotency/stemness. On the basis of the observations reported in these studies, we draw attention to potential molecular connections between extra-telomeric biology and pluripotency. Finally, in the context of the nonlocal influence of telomeres on pluripotency and stemness, we discuss major opportunities for progress in molecular understanding of aging-related disorders and neurodegenerative diseases.
Keywords: chromosome end; de-differentiation; extra-telomeric function; gene regulation; genome organization; neurodegenerative disease; pluripotency; shelterin; stem cells; telomere.
© 2020 Vinayagamurthy et al.
Conflict of interest statement
Conflict of interest—The authors declare that they have no conflicts of interest with the contents of this article.
Figures


Similar articles
-
Telomeres expand sphere of influence: emerging molecular impact of telomeres in non-telomeric functions.Trends Genet. 2023 Jan;39(1):59-73. doi: 10.1016/j.tig.2022.10.002. Epub 2022 Nov 17. Trends Genet. 2023. PMID: 36404192 Free PMC article. Review.
-
Telomere length-dependent transcription and epigenetic modifications in promoters remote from telomere ends.PLoS Genet. 2018 Nov 15;14(11):e1007782. doi: 10.1371/journal.pgen.1007782. eCollection 2018 Nov. PLoS Genet. 2018. PMID: 30439955 Free PMC article.
-
TRF2-independent chromosome end protection during pluripotency.Nature. 2021 Jan;589(7840):103-109. doi: 10.1038/s41586-020-2960-y. Epub 2020 Nov 25. Nature. 2021. PMID: 33239783 Free PMC article.
-
Telomere repeat-binding factor 2 binds extensively to extra-telomeric G-quadruplexes and regulates the epigenetic status of several gene promoters.J Biol Chem. 2019 Nov 22;294(47):17709-17722. doi: 10.1074/jbc.RA119.008687. Epub 2019 Oct 1. J Biol Chem. 2019. PMID: 31575660 Free PMC article.
-
The Intra- and Extra-Telomeric Role of TRF2 in the DNA Damage Response.Int J Mol Sci. 2021 Sep 14;22(18):9900. doi: 10.3390/ijms22189900. Int J Mol Sci. 2021. PMID: 34576063 Free PMC article. Review.
Cited by
-
Telomere length sensitive regulation of interleukin receptor 1 type 1 (IL1R1) by the shelterin protein TRF2 modulates immune signalling in the tumour microenvironment.Elife. 2024 Dec 27;13:RP95106. doi: 10.7554/eLife.95106. Elife. 2024. PMID: 39728924 Free PMC article.
-
Physical Activity on Telomere Length as a Biomarker for Aging: A Systematic Review.Sports Med Open. 2022 Sep 4;8(1):111. doi: 10.1186/s40798-022-00503-1. Sports Med Open. 2022. PMID: 36057868 Free PMC article.
-
Human telomerase is directly regulated by non-telomeric TRF2-G-quadruplex interaction.Cell Rep. 2021 May 18;35(7):109154. doi: 10.1016/j.celrep.2021.109154. Cell Rep. 2021. PMID: 34010660 Free PMC article.
-
Telomere length in leucocytes and solid tissues of young and aged rats.Aging (Albany NY). 2022 Feb 27;14(4):1713-1728. doi: 10.18632/aging.203922. Epub 2022 Feb 27. Aging (Albany NY). 2022. PMID: 35220278 Free PMC article.
-
Telomeres expand sphere of influence: emerging molecular impact of telomeres in non-telomeric functions.Trends Genet. 2023 Jan;39(1):59-73. doi: 10.1016/j.tig.2022.10.002. Epub 2022 Nov 17. Trends Genet. 2023. PMID: 36404192 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

