Activation of allylic esters in an intramolecular vinylogous kinetic resolution reaction with synergistic magnesium catalysts
- PMID: 32444612
- PMCID: PMC7244749
- DOI: 10.1038/s41467-020-16486-0
Activation of allylic esters in an intramolecular vinylogous kinetic resolution reaction with synergistic magnesium catalysts
Abstract
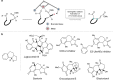
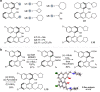
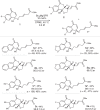
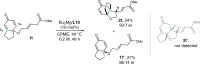
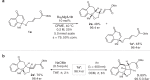
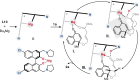
Kinetic resolution (KR) of racemic starting materials is a powerful and practical alternative to prepare valuable enantiomerically enriched compounds. A magnesium-catalyzed kinetic resolution based on a designed intramolecular vinylogous Michael reaction is disclosed. Here we show a synergistic catalytic strategy based on the development of chiral ligands. Substrates containing linear allylic ester structures are designed and synthesized to construct key [6.6.5]-tricyclic chiral skeletons via this kinetic resolution process. Detailed mechanistic studies reveal a rational mechanism for the current intramolecular vinylogous KR reaction. The desired direct intramolecular asymmetric vinylogous Michael reaction of linear allylic esters is realized in high efficiency and enantioselectivity with the synergistic catalytic system.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Applications of Iridium-Catalyzed Asymmetric Allylic Substitution Reactions in Target-Oriented Synthesis.Acc Chem Res. 2017 Oct 17;50(10):2539-2555. doi: 10.1021/acs.accounts.7b00300. Epub 2017 Sep 22. Acc Chem Res. 2017. PMID: 28937739
-
Desymmetrization Process by Mg(II)-Catalyzed Intramolecular Vinylogous Michael Reaction.Org Lett. 2020 Dec 4;22(23):9229-9233. doi: 10.1021/acs.orglett.0c03417. Epub 2020 Nov 17. Org Lett. 2020. PMID: 33201719
-
Catalytic Asymmetric Conjugate Addition of 2-Methyl-3,5-dinitrobenzoates to Unsaturated Ketones.J Org Chem. 2022 May 6;87(9):5497-5509. doi: 10.1021/acs.joc.1c02757. Epub 2022 Apr 14. J Org Chem. 2022. PMID: 35420811
-
Copper-catalyzed divergent kinetic resolution of racemic allylic substrates.Chirality. 2011 Oct;23(9):703-10. doi: 10.1002/chir.20980. Epub 2011 Aug 12. Chirality. 2011. PMID: 21837639 Review.
-
Advances in asymmetric oxidative kinetic resolution of racemic secondary alcohols catalyzed by chiral Mn(III) salen complexes.Chirality. 2017 Dec;29(12):798-810. doi: 10.1002/chir.22768. Epub 2017 Sep 29. Chirality. 2017. PMID: 28963733 Review.
Cited by
-
Harnessing Dpp-Imine as a Powerful Achiral Cocatalyst to Dramatically Increase the Efficiency and Stereoselectivity in a Magnesium-Mediated Oxa-Michael Reaction.JACS Au. 2023 Dec 21;4(1):164-176. doi: 10.1021/jacsau.3c00584. eCollection 2024 Jan 22. JACS Au. 2023. PMID: 38274262 Free PMC article.
References
-
- Keith JM, Larrow JF, Jacobsen EN. Practical considerations in kinetic resolution reactions. Adv. Synth. Catal. 2001;343:5–26.
-
- Robinson DEJE, Bull SD. Kinetic resolution strategies using non-enzymatic catalysts. Tetrahedron: Asymmetry. 2003;14:1407–1446.
-
- Vedejs E, Jure M. Efficiency in nonenzymatic kinetic resolution. Angew. Chem. Int. Ed. 2005;44:3974–4001. - PubMed
-
- Pellissier H. Catalytic non‐enzymatic kinetic resolution. Adv. Synth. Catal. 2011;353:1613–1666.
-
- La DS, et al. Mo-catalyzed asymmetric synthesis of dihydrofurans. catalytic kinetic resolution and enantioselective desymmetrization through ring-closing metathesis. J. Am. Chem. Soc. 1998;120:9720–9721.
Publication types
LinkOut - more resources
Full Text Sources

