The Role of Mitochondrial Impairment in Alzheimer´s Disease Neurodegeneration: The Tau Connection
- PMID: 32448104
- PMCID: PMC7709157
- DOI: 10.2174/1570159X18666200525020259
The Role of Mitochondrial Impairment in Alzheimer´s Disease Neurodegeneration: The Tau Connection
Abstract
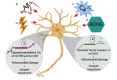
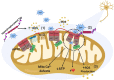
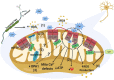
Accumulative evidence has shown that mitochondrial dysfunction plays a pivotal role in the pathogenesis of Alzheimer's disease (AD). Mitochondrial impairment actively contributes to the synaptic and cognitive failure that characterizes AD. The presence of soluble pathological forms of tau like hyperphosphorylated at Ser396 and Ser404 and cleaved at Asp421 by caspase 3, negatively impacts mitochondrial bioenergetics, transport, and morphology in neurons. These adverse effects against mitochondria health will contribute to the synaptic impairment and cognitive decline in AD. Current studies suggest that mitochondrial failure induced by pathological tau forms is likely the result of the opening of the mitochondrial permeability transition pore (mPTP). mPTP is a mitochondrial mega-channel that is activated by increases in calcium and is associated with mitochondrial stress and apoptosis. This structure is composed of different proteins, where Ciclophilin D (CypD) is considered to be the primary mediator of mPTP activation. Also, new studies suggest that mPTP contributes to Aβ pathology and oxidative stress in AD. Further, inhibition of mPTP through the reduction of CypD expression prevents cognitive and synaptic impairment in AD mouse models. More importantly, tau protein contributes to the physiological regulation of mitochondria through the opening/interaction with mPTP in hippocampal neurons. Therefore, in this paper, we will discuss evidence that suggests an important role of pathological forms of tau against mitochondrial health. Also, we will discuss the possible role of mPTP in the mitochondrial impairment produced by the presence of tau pathology and its impact on synaptic function present in AD.
Keywords: Alzheimer´s disease; Mitochondria; Tau; calcium; mitochondrial permeability transition pore; oxidative stress.
Copyright© Bentham Science Publishers; For any queries, please email at epub@benthamscience.net.
Figures
Similar articles
-
Cyclophilin D (CypD) ablation prevents neurodegeneration and cognitive damage induced by caspase-3 cleaved tau.Free Radic Biol Med. 2025 May;232:128-141. doi: 10.1016/j.freeradbiomed.2025.02.035. Epub 2025 Feb 27. Free Radic Biol Med. 2025. PMID: 40023297
-
Caspase-3 cleaved tau impairs mitochondrial function through the opening of the mitochondrial permeability transition pore.Biochim Biophys Acta Mol Basis Dis. 2024 Jan;1870(1):166898. doi: 10.1016/j.bbadis.2023.166898. Epub 2023 Sep 28. Biochim Biophys Acta Mol Basis Dis. 2024. PMID: 37774936 Free PMC article.
-
Mitochondrial Calcium Deregulation in the Mechanism of Beta-Amyloid and Tau Pathology.Cells. 2020 Sep 21;9(9):2135. doi: 10.3390/cells9092135. Cells. 2020. PMID: 32967303 Free PMC article. Review.
-
Mitochondria-tau association promotes cognitive decline and hippocampal bioenergetic deficits during the aging.Free Radic Biol Med. 2024 May 1;217:141-156. doi: 10.1016/j.freeradbiomed.2024.03.017. Epub 2024 Mar 27. Free Radic Biol Med. 2024. PMID: 38552927
-
Synaptic Mitochondria: An Early Target of Amyloid-β and Tau in Alzheimer's Disease.J Alzheimers Dis. 2021;84(4):1391-1414. doi: 10.3233/JAD-215139. J Alzheimers Dis. 2021. PMID: 34719499 Review.
Cited by
-
In-vitro and in-vivo studies of two-drug cocktail therapy targeting chemobrain via the Nrf2/NF-κB signaling pathway.J Mol Histol. 2024 Aug;55(4):599-625. doi: 10.1007/s10735-024-10217-1. Epub 2024 Jul 23. J Mol Histol. 2024. PMID: 39042217
-
mTOR signaling and Alzheimer's disease: What we know and where we are?CNS Neurosci Ther. 2024 Apr;30(4):e14463. doi: 10.1111/cns.14463. Epub 2023 Sep 18. CNS Neurosci Ther. 2024. PMID: 37721413 Free PMC article. Review.
-
Lysophosphatidylcholines are associated with P-tau181 levels in early stages of Alzheimer's Disease.Res Sq [Preprint]. 2024 Jan 8:rs.3.rs-3346076. doi: 10.21203/rs.3.rs-3346076/v1. Res Sq. 2024. PMID: 38260644 Free PMC article. Preprint.
-
Role of Calcium Modulation in the Pathophysiology and Treatment of Alzheimer's Disease.Int J Mol Sci. 2023 May 22;24(10):9067. doi: 10.3390/ijms24109067. Int J Mol Sci. 2023. PMID: 37240413 Free PMC article. Review.
-
Melatonin Ameliorates Post-Stroke Cognitive Impairment in Mice by Inhibiting Excessive Mitophagy.Cells. 2024 May 18;13(10):872. doi: 10.3390/cells13100872. Cells. 2024. PMID: 38786094 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

