Real-time monitoring of bacterial biofilms metabolic activity by a redox-reactive nanosensors array
- PMID: 32448291
- PMCID: PMC7247256
- DOI: 10.1186/s12951-020-00637-y
Real-time monitoring of bacterial biofilms metabolic activity by a redox-reactive nanosensors array
Abstract
Background: Bacterial biofilms are communities of surface-associated microorganisms living in cellular clusters or micro-colonies, encapsulated in a complex matrix composed of an extracellular polymeric substance, separated by open water channels that act as a circulatory system that enable better diffusion of nutrients and easier removal of metabolic waste products. The monitoring of biofilms can provide important information on fundamental biofilm-related processes. That information can shed light on the bacterial processes and enable scientists to find ways of preventing future bacterial infections. Various approaches in use for biofilm analysis are based on microscopic, spectrochemical, electrochemical, and piezoelectrical methods. All these methods provide significant progress in understanding the bio-process related to biofilm formation and eradication, nevertheless, the development of novel approaches for the real-time monitoring of biochemical, in particular metabolic activity, of bacterial species during the formation, life and eradication of biofilms is of great potential importance.
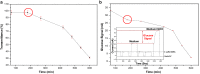
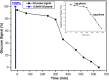
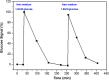
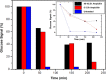
Results: Here, detection and monitoring of the metabolic activity of bacterial biofilms in high-ionic-strength solutions were enabled as a result of novel surface modification by an active redox system, composed of 9,10-dihydroxyanthracene/9,10-anthraquinone, on the oxide layer of the SiNW, yielding a chemically-gated FET array. With the use of enzymatic reactions of oxidases, metabolites can be converted to H2O2 and monitored by the nanosensors. Here, the successful detection of glucose metabolites in high-ionic-strength solutions, such as bacterial media, without pre-processing of small volume samples under different conditions and treatments, has been demonstrated. The biofilms were treated with antibiotics differing in their mechanisms of action and were compared to untreated biofilms. Further examination of biofilms under antibiotic treatment with SiNW-FET devices could shed light on the bioprocess that occurs within the biofilm. Moreover, finding proper treatment that eliminates the biofilm could be examined by the novel nanosensor as a monitoring tool.
Conclusions: To summarize, the combination of redox-reactive SiNW-FET devices with micro-fluidic techniques enables the performance of rapid, automated, and real-time metabolite detection with the use of minimal sample size, noninvasively and label-free. This novel platform can be used as an extremely sensitive tool for detection and establishing medical solutions for bacterial-biofilm eradication and for finding a proper treatment to eliminate biofilm contaminations. Moreover, the sensing system can be used as a research tool for further understanding of the metabolic processes that occur within the bacterial biofilm population.
Keywords: Bacteria; Biofilms; Field effect transistors; Metabolism; Nanosensors; Silicon nanowires.
Figures


Similar articles
-
Redox-Reactive Field-Effect Transistor Nanodevices for the Direct Monitoring of Small Metabolites in Biofluids toward Implantable Nanosensors Arrays.ACS Nano. 2020 Mar 24;14(3):3587-3594. doi: 10.1021/acsnano.9b10090. Epub 2020 Mar 9. ACS Nano. 2020. PMID: 32129979 Free PMC article.
-
Silicon nanowires as field-effect transducers for biosensor development: a review.Anal Chim Acta. 2014 May 12;825:1-25. doi: 10.1016/j.aca.2014.03.016. Epub 2014 Mar 15. Anal Chim Acta. 2014. PMID: 24767146 Review.
-
Cellular Metabolomics by a Universal Redox-Reactive Nanosensors Array: From the Cell Level to Tumor-on-a-Chip Analysis.Nano Lett. 2019 Apr 10;19(4):2478-2488. doi: 10.1021/acs.nanolett.9b00052. Epub 2019 Mar 22. Nano Lett. 2019. PMID: 30884235
-
Silicon nanowire arrays for label-free detection of DNA.Anal Chem. 2007 May 1;79(9):3291-7. doi: 10.1021/ac061808q. Epub 2007 Apr 4. Anal Chem. 2007. PMID: 17407259
-
Silicon nanowire field-effect-transistor based biosensors: from sensitive to ultra-sensitive.Biosens Bioelectron. 2014 Oct 15;60:101-11. doi: 10.1016/j.bios.2014.03.057. Epub 2014 Apr 15. Biosens Bioelectron. 2014. PMID: 24787124 Review.
Cited by
-
Depletion of Highly Abundant Protein Species from Biosamples by the Use of a Branched Silicon Nanopillar On-Chip Platform.Anal Chem. 2021 Nov 2;93(43):14527-14536. doi: 10.1021/acs.analchem.1c03506. Epub 2021 Oct 20. Anal Chem. 2021. PMID: 34668374 Free PMC article.
-
Influence of surface characteristics of implant materials on MRSA biofilm formation and effects of antimicrobial treatment.Front Microbiol. 2023 Apr 20;14:1145210. doi: 10.3389/fmicb.2023.1145210. eCollection 2023. Front Microbiol. 2023. PMID: 37152752 Free PMC article.
-
Microbial Electrochemical Systems: Principles, Construction and Biosensing Applications.Sensors (Basel). 2021 Feb 11;21(4):1279. doi: 10.3390/s21041279. Sensors (Basel). 2021. PMID: 33670122 Free PMC article. Review.
-
Understanding bacterial biofilms: From definition to treatment strategies.Front Cell Infect Microbiol. 2023 Apr 6;13:1137947. doi: 10.3389/fcimb.2023.1137947. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 37091673 Free PMC article.
-
The "Bloodless" Blood Test: Intradermal Prick Nanoelectronics for the Blood Extraction-Free Multiplex Detection of Protein Biomarkers.ACS Nano. 2022 Sep 27;16(9):13800-13813. doi: 10.1021/acsnano.2c01793. Epub 2022 Aug 25. ACS Nano. 2022. PMID: 36006419 Free PMC article.
References
-
- Cui Y, Duan X, Hu J, Lieber CM. Doping and electrical transport in silicon nanowires. J Phys Chem B. 2000;104:5213–5216.
-
- Xia Y, Yang P, Sun Y, Wu Y, Mayers B, Gates B, Yin Y, Kim F, Yan H. One-dimensional nanostructures: synthesis, characterization, and applications. Adv Mater. 2003;15:353–389.
-
- Peretz-Soroka H, Pevzner A, Davidi G, Naddaka V, Tirosh R, Flaxer E, Patolsky F. Optically-gated self-calibrating nanosensors: monitoring pH and metabolic activity of living cells. Nano Lett. 2013;13:3157–3168. - PubMed
-
- Kwiat M, Elnathan R, Kwak M, de Vries JW, Pevzner A, Engel Y, Burstein L, Khatchtourints A, Lichtenstein A, Flaxer E. Non-covalent monolayer-piercing anchoring of lipophilic nucleic acids: preparation, characterization, and sensing applications. J Am Chem Soc. 2011;134:280–292. - PubMed
-
- Pevzner A, Engel Y, Elnathan R, Ducobni T, Ben-Ishai M, Reddy K, Shpaisman N, Tsukernik A, Oksman M, Patolsky F. Knocking down highly-ordered large-scale nanowire arrays. Nano Lett. 2010;10:1202–1208. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

