The cysteine protease ATG4B of Trichinella spiralis promotes larval invasion into the intestine of the host
- PMID: 32448368
- PMCID: PMC7245929
- DOI: 10.1186/s13567-020-00791-z
The cysteine protease ATG4B of Trichinella spiralis promotes larval invasion into the intestine of the host
Abstract
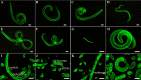
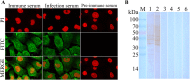
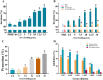
The cysteine proteases of parasites are vital contributors that induce parasite migration to and invasion of host tissue. In this study, we analysed the cysteine protease ATG4B of Trichinella spiralis (TsATG4B) isolated from the soluble proteins of Trichinella spiralis (T. spiralis) adult worms to ascertain its biochemical properties and functions during invasion into the intestine of the host. The 43 kDa recombinant cysteine protease ATG4B protein (rTsATG4B) consists of a conserved peptidase_C54 domain and was expressed in Escherichia coli. Gelatine zymography showed that rTsATG4B could hydrolyse gelatine and that the hydrolytic activity was prevented by the cysteine protease inhibitor E-64 (pH 5.2). Immunofluorescence assays showed that TsATG4B is expressed at different stages and is localized at the cuticles and stichosomes of worms. Far-Western blotting and confocal microscopy revealed that rTsATG4B interacts with intestinal epithelial cells (IECs) and that it was subcellularly localized to the membrane and cytoplasm in IECs. Real‑time quantitative PCR (qPCR) results indicated that the transcription level of the TsATG4B gene was the higher in 6-day-old adult worms (6 days AW) than in any other stage. An in vitro larval invasion assay verified that rTsATG4B promoted larval invasion and that invasion was inhibited when rTsATG4B was pre-incubated with E-64, whereas anti-rTsATG4B serum inhibited larval invasion in a dose-dependent manner. Collectively, these results suggested that the enzymatic activity of TsATG4B significantly influences the hydrolysis process, which is necessary for larval invasion of the host intestinal epithelium.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures





Similar articles
-
Characterization of a novel cysteine protease in Trichinella spiralis and its role in larval intrusion, development and fecundity.Vet Res. 2021 Aug 26;52(1):113. doi: 10.1186/s13567-021-00983-1. Vet Res. 2021. PMID: 34446106 Free PMC article.
-
Cloning, expression and characterisation of a cysteine protease from Trichinella spiralis.Folia Parasitol (Praha). 2018 May 29;65:2018.007. doi: 10.14411/fp.2018.007. Folia Parasitol (Praha). 2018. PMID: 29905572
-
Characterization of a serine protease inhibitor from Trichinella spiralis and its participation in larval invasion of host's intestinal epithelial cells.Parasit Vectors. 2018 Sep 6;11(1):499. doi: 10.1186/s13071-018-3074-3. Parasit Vectors. 2018. PMID: 30189888 Free PMC article.
-
Trichinella spiralis and the concept of niche.J Parasitol. 1993 Aug;79(4):472-82. J Parasitol. 1993. PMID: 8331468 Review.
-
Invasion of epithelial cells by Trichinella spiralis: in vitro observations.Parasite. 2001 Jun;8(2 Suppl):S48-50. doi: 10.1051/parasite/200108s2048. Parasite. 2001. PMID: 11484381 Review.
Cited by
-
Proteases secreted by Trichinella spiralis intestinal infective larvae damage the junctions of the intestinal epithelial cell monolayer and mediate larval invasion.Vet Res. 2022 Mar 7;53(1):19. doi: 10.1186/s13567-022-01032-1. Vet Res. 2022. PMID: 35255974 Free PMC article.
-
Characterization of a novel cysteine protease in Trichinella spiralis and its role in larval intrusion, development and fecundity.Vet Res. 2021 Aug 26;52(1):113. doi: 10.1186/s13567-021-00983-1. Vet Res. 2021. PMID: 34446106 Free PMC article.
-
Plancitoxin-1 mediates extracellular trap evasion by the parasitic helminth Trichinella spiralis.BMC Biol. 2024 Jul 29;22(1):158. doi: 10.1186/s12915-024-01958-2. BMC Biol. 2024. PMID: 39075478 Free PMC article.
-
Inhibition of GSDMD-mediated pyroptosis triggered by Trichinella spiralis intervention contributes to the alleviation of DSS-induced ulcerative colitis in mice.Parasit Vectors. 2023 Aug 14;16(1):280. doi: 10.1186/s13071-023-05857-3. Parasit Vectors. 2023. PMID: 37580819 Free PMC article.
References
-
- Murrell KD. The dynamics of Trichinella spiralis epidemiology: out to pasture? Vet Parasitol. 2016;231:92–96. - PubMed
-
- Cui J, Wang Z, Xu B. The epidemiology of human trichinellosis in China during 2004–2009. Acta Trop. 2011;118:1–5. - PubMed
-
- Rostami A, Gamble HR, Dupouy-Camet J, Khazan H, Bruschi F. Meat sources of infection for outbreaks of human trichinellosis. Food Microbiol. 2017;64:65–71. - PubMed
-
- Jiang P, Zhang X, Wang LA, Han LH, Yang M, Duan JY, Sun GG, Qi X, Liu RD, Wang ZQ. Survey of Trichinella infection from domestic pigs in the historical endemic areas of Henan province, central China. Parasitol Res. 2016;115:4707–4709. - PubMed
-
- Sequeira GJ, Zbrun MV, Soto LP, Astesana DM, Blajman JE, Rosmini MR, Frizzo LS, Signorini M. Quantitative risk assessment of human trichinellosis caused by consumption of pork meat sausages in Argentina. Zoonoses Public Health. 2016;63:167–176. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

