Insights into Substrate and Inhibitor Selectivity among Human GLUT Transporters through Comparative Modeling and Molecular Docking
- PMID: 32462103
- PMCID: PMC7244221
- DOI: 10.1021/acsomega.8b03447
Insights into Substrate and Inhibitor Selectivity among Human GLUT Transporters through Comparative Modeling and Molecular Docking
Abstract
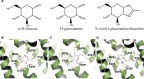
The solute carrier 2 family is composed of 14 transporters, which are members of the major facilitator superfamily. Despite their high physiological importance, there are still many open questions concerning their function and specificity, and in some cases, their physiological substrate is still unknown. To understand the determinants of the substrate and inhibitor specificity, we modeled all human glucose transport carriers (GLUTs) and simulated their interaction with known ligands. Comparative modeling was performed with the @TOME-2 pipeline, employing multiple templates and providing an ensemble of models for each GLUT. We analyzed models in both outward-occluded and inward-open conformations, to compare exofacial and endofacial binding sites throughout the family and understand differences in susceptibility of GLUTs to the inhibitor cytochalasin B. Finally, we employed molecular docking and bioinformatics to identify residues likely critical for recognition of myo-inositol by GLUT13 and urate by GLUT9. These results provide insights into the molecular basis for the specificity for these substrates. In addition, we suggested a potential recognition site of glucosamine by GLUT11 to be evaluated in future experiments.
Copyright © 2019 American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




References
LinkOut - more resources
Full Text Sources
