Production and characterization of a monoclonal antibody against the sialidase of Gardnerella vaginalis using a synthetic peptide in a MAP8 format
- PMID: 32462244
- PMCID: PMC7253150
- DOI: 10.1007/s00253-020-10691-z
Production and characterization of a monoclonal antibody against the sialidase of Gardnerella vaginalis using a synthetic peptide in a MAP8 format
Abstract
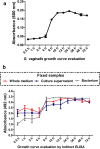
Bacterial vaginosis is one of the most frequent vaginal infections. Its main etiological agent is Gardnerella vaginalis, which produces several virulence factors involved in vaginal infection and colonization, in particular, sialidase (SLD), a potential clinical biomarker that participates in immune response modulation and mucus degradation. The main objective of this work was the production and evaluation of a monoclonal antibody against G. vaginalis sialidase and its validation in immunoassays. For immunization of mice, a synthetic multiantigenic peptide was used, and hybridomas were generated. After fusion, hybridomas were evaluated for antibody production and cloned by limited dilution. One clone producing IgG1 was selected and characterized by indirect ELISA, dot blot, and Western blot, and we also tested clinical isolates and HeLa cells infected with G. vaginalis. The results showed that the anti-SLD antibody recognized a single protein of ~90 kDa that correlated with the estimated molecular weight of SLD. In addition, anti-SLD antibody recognized SLD from complete bacteria and from culture supernatants of infected Hela cells. In conclusion, our results showed that the anti-SLD antibody recognized SLD from different sources and could be considered a new tool for the diagnosis of bacterial vaginosis. KEY POINTS: • Anti-sialidase mAb was generated using a synthetic peptide • The mAb recognizes synthetic peptide and intact protein from multiple sources • The antibody was characterized by several immunological methods.
Keywords: Bacterial vaginosis; Gardnerella vaginalis; Monoclonal antibody; Sialidase.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures





References
-
- Bautista CT, Wurapa E, Sateren WB, Morris S, Hollingsworth B, Sanchez JL. Bacterial vaginosis: a synthesis of the literature on etiology, prevalence, risk factors, and relationship with chlamydia and gonorrhea infections. Mil Med Res. 2016;3:1–10. doi: 10.1186/s40779-016-0074-5. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

