Determining the optimal dosing of a novel combination regimen of ceftazidime/avibactam with aztreonam against NDM-1-producing Enterobacteriaceae using a hollow-fibre infection model
- PMID: 32464664
- PMCID: PMC8444334
- DOI: 10.1093/jac/dkaa197
Determining the optimal dosing of a novel combination regimen of ceftazidime/avibactam with aztreonam against NDM-1-producing Enterobacteriaceae using a hollow-fibre infection model
Abstract
Background: MBL-producing strains of Enterobacteriaceae are a major public health concern. We sought to define optimal combination regimens of ceftazidime/avibactam with aztreonam in a hollow-fibre infection model (HFIM) of MBL-producing strains of Escherichia coli and Klebsiella pneumoniae.
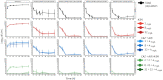
Methods: E. coli ARLG-1013 (blaNDM-1, blaCTX-M, blaCMY, blaTEM) and K. pneumoniae ARLG-1002 (blaNDM-1, blaCTXM-15, blaDHA, blaSHV, blaTEM) were studied in the HFIM using simulated human dosing regimens of ceftazidime/avibactam and aztreonam. Experiments were designed to evaluate the effect of staggered versus simultaneous administration, infusion duration and aztreonam daily dose (6 g/day versus 8 g/day) on bacterial killing and resistance suppression. Prospective validation experiments for the most active combination regimens were performed in triplicate to ensure reproducibility.
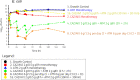
Results: Staggered administration of the combination (ceftazidime/avibactam followed by aztreonam) was found to be inferior to simultaneous administration. Longer infusion durations (2 h and continuous infusion) also resulted in enhanced bacterial killing relative to 30 min infusions. The rate of killing was more pronounced with 8 g/day versus 6 g/day aztreonam combination regimens for both tested strains. In the prospective validation experiments, ceftazidime/avibactam with aztreonam dosed every 8 and 6 h, respectively (ceftazidime/avibactam 2/0.5 g every 8 h + aztreonam 2 g every 6 h), or ceftazidime/avibactam with aztreonam as continuous infusions resulted in maximal bacterial killing and resistance suppression over 7 days.
Conclusions: Simultaneous administration of aztreonam 8 g/day given as a continuous or 2 h infusion with ceftazidime/avibactam resulted in complete bacterial eradication and resistance suppression. Further study of this combination is needed with additional MBL-producing Gram-negative pathogens. The safety of this double β-lactam strategy also warrants further study in Phase 1 clinical trials.
© The Author(s) 2020. Published by Oxford University Press on behalf of the British Society for Antimicrobial Chemotherapy. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Figures



References
-
- IDSA. Antibacterial Resistance. https://www.idsociety.org/policy--advocacy/antimicrobial-resistance/.
-
- Wright H, Bonomo RA, Paterson DL.. New agents for the treatment of infections with Gram-negative bacteria: restoring the miracle or false dawn? Clin Microbiol Infect 2017; 23: 704–12. - PubMed
-
- Sader HS, Castanheira M, Duncan LR. et al.Antimicrobial susceptibility of Enterobacteriaceae and Pseudomonas aeruginosa isolates from United States medical centers stratified by infection type: results from the International Network for Optimal Resistance Monitoring (INFORM) Surveillance Program, 2015-2016. Diagn Microbiol Infect Dis 2018; 92: 69–74. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

