Use of Thrombodynamics for revealing the participation of platelet, erythrocyte, endothelial, and monocyte microparticles in coagulation activation and propagation
- PMID: 32469873
- PMCID: PMC7259734
- DOI: 10.1371/journal.pone.0227932
Use of Thrombodynamics for revealing the participation of platelet, erythrocyte, endothelial, and monocyte microparticles in coagulation activation and propagation
Abstract
Background and objective: For many pathological states, microparticles are supposed to be one of the causes of hypercoagulation. Although there are some indirect data about microparticles participation in coagulation activation and propagation, the integral hemostasis test Thrombodynamics allows to measure micropaticles participation in these two coagulation phases directly. Demonstrates microparticles participation in coagulation activation by influence on the appearance of coagulation centres in the plasma volume and the rate of clot growth from the surface with immobilized tissue factor.Methods: Microparticles were obtained from platelets and erythrocytes by stimulation with thrombin receptor-activating peptide (SFLLRN) and calcium ionophore (A23187), respectively, from monocytes, endothelial HUVEC culture and monocytic THP cell culture by stimulation with lipopolysaccharides. Microparticles were counted by flow cytometry and titrated in microparticle-depleted normal plasma in the Thrombodynamics test.
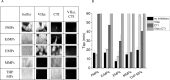
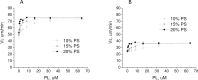
Results: Monocyte microparticles induced the appearance of clotting centres through the TF pathway at concentrations approximately 100-fold lower than platelet and erythrocyte microparticles, which activated plasma by the contact pathway. For endothelial microparticles, both activation pathways were essential, and their activity was intermediate. Monocyte microparticles induced plasma clotting by the appearance of hundreds of clots with an extremely slow growth rate, while erythrocyte microparticles induced the appearance of a few clots with a growth rate similar to that from surface covered with high-density tissue factor. Patterns of clotting induced by platelet and endothelial microparticles were intermediate. Platelet, erythrocyte and endothelial microparticles impacts on the rate of clot growth from the surface with tissue factor did not differ significantly within the 0-200·103/ul range of microparticles concentrations. However, at concentrations greater than 500·103/ul, erythrocyte microparticles increased the stationary clot growth rate to significantly higher levels than do platelet microparticles or artificial phospholipid vesicles consisting of phosphatidylcholine and phosphatidylserine.
Conclusion: Microparticles of different origins demonstrated qualitatively different characteristics related to coagulation activation and propagation.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
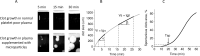






References
-
- Berckmans RJ, Nieuwland R, Boing AN, Romijn FP, Hack CE, Sturk A. Cell-derived microparticles circulate in healthy humans and support low grade thrombin generation. Thromb Haemost. 2001;85: 639–646. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

