Central Airway Toxicity After High Dose Radiation: A Combined Analysis of Prospective Clinical Trials for Non-Small Cell Lung Cancer
- PMID: 32470501
- PMCID: PMC8074530
- DOI: 10.1016/j.ijrobp.2020.05.026
Central Airway Toxicity After High Dose Radiation: A Combined Analysis of Prospective Clinical Trials for Non-Small Cell Lung Cancer
Abstract
Purpose: To study the dosimetric risk factors for radiation-induced proximal bronchial tree (PBT) toxicity in patients treated with radiation therapy for non-small cell lung cancer (NSCLC).
Methods and materials: Patients with medically inoperable or unresectable NSCLC treated with conventionally fractionated 3-dimensional conformal radiation therapy (3DCRT) in prospective clinical trials were eligible for this study. Proximal bronchial tree (PBT) and PBT wall were contoured consistently per RTOG 1106 OAR-Atlas. The dose-volume histograms (DVHs) of physical prescription dose (DVHp) and biological effective dose (α/β = 2.5; DVH2.5) were generated, respectively. The primary endpoint was PBT toxicities, defined by CTCAE 4.0 under the terminology of bronchial stricture/atelectasis.
Results: Of 100 patients enrolled, with a median follow-up of 64 months (95% confidence interval [CI], 50-78), 73% received 70 Gy or greater and 17% developed PBT toxicity (grade 1, 8%; grade 2, 6%; grade 3, 0%; and grade 4, 3%). The median time interval between RT initiation and onset of PBT toxicity was 8.4 months (95% CI, 4.7-44.1). The combined DVHs showed that no patient with a PBT maximum physical dose <65 Gy developed any PBT toxicity. Cox proportional hazards analysis and receiver operating characteristic analysis demonstrated that V75 of PBT was the most significant dosimetric parameter for both grade 1+ (P = .035) and grade 2+ (P = .037) PBT toxicities. The dosimetric thresholds for V75 of PBT were 6.8% and 11.9% for grade 1+ and grade 2+ PBT toxicity, respectively.
Conclusions: V75 of PBT appeared be the most significant dosimetric parameter for PBT toxicity after conventionally fractionated thoracic 3DCRT. Constraining V75 of PBT can limit clinically significant PBT toxicity.
Copyright © 2020. Published by Elsevier Inc.
Conflict of interest statement
【Conflict of Interest】
The authors declare no potential conflict of interest related to this work.
Figures



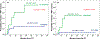
Similar articles
-
Validation of RTOG 0813 Proximal Bronchial Tree Constraints for Pulmonary Toxicity With Stereotactic Body Radiation Therapy for Central Non-small Cell Lung Cancer.Int J Radiat Oncol Biol Phys. 2020 May 1;107(1):72-78. doi: 10.1016/j.ijrobp.2020.01.009. Epub 2020 Jan 25. Int J Radiat Oncol Biol Phys. 2020. PMID: 31987965 Clinical Trial.
-
Dosimetric Factors and Toxicity in Highly Conformal Thoracic Reirradiation.Int J Radiat Oncol Biol Phys. 2016 Mar 15;94(4):808-15. doi: 10.1016/j.ijrobp.2015.12.007. Epub 2015 Dec 17. Int J Radiat Oncol Biol Phys. 2016. PMID: 26831903
-
Clinical and dosimetric predictors of radiation-induced esophageal toxicity.Int J Radiat Oncol Biol Phys. 1999 Aug 1;45(1):97-103. doi: 10.1016/s0360-3016(99)00163-7. Int J Radiat Oncol Biol Phys. 1999. PMID: 10477012
-
Dosimetric and clinical predictors for radiation-induced esophageal injury.Int J Radiat Oncol Biol Phys. 2005 Feb 1;61(2):335-47. doi: 10.1016/j.ijrobp.2004.06.014. Int J Radiat Oncol Biol Phys. 2005. PMID: 15667951 Review.
-
Medical consequences of radiation exposure of the bronchi-what can we learn from high-dose precision radiation therapy?J Radiol Prot. 2021 Nov 3;41(4). doi: 10.1088/1361-6498/ac28ef. J Radiol Prot. 2021. PMID: 34547741 Review.
Cited by
-
Value of carbon-ion radiotherapy for early stage non-small cell lung cancer.Clin Transl Radiat Oncol. 2022 Jun 14;36:16-23. doi: 10.1016/j.ctro.2022.06.005. eCollection 2022 Sep. Clin Transl Radiat Oncol. 2022. PMID: 35756194 Free PMC article. Review.
-
Central airway squamous metaplasia following radiation therapy mimicking local tumour recurrence.Respir Med Case Rep. 2023 Nov 2;46:101942. doi: 10.1016/j.rmcr.2023.101942. eCollection 2023. Respir Med Case Rep. 2023. PMID: 38025247 Free PMC article.
-
Are treatment plans optimized on the basis of acuros XB dose calculation robust against anatomic changes during online adaptive radiotherapy for lung cancer regarding dose homogeneity?Radiat Oncol. 2025 May 15;20(1):75. doi: 10.1186/s13014-025-02656-1. Radiat Oncol. 2025. PMID: 40375212 Free PMC article.
-
Central airway squamous metaplasia following radiation therapy mimicking local tumour recurrence.Respir Med Case Rep. 2023 Nov 7;46:101945. doi: 10.1016/j.rmcr.2023.101945. eCollection 2023. Respir Med Case Rep. 2023. PMID: 38074083 Free PMC article.
-
SPECT/CT Dosimetry of Bronchial Artery 99mTc Macroaggregated Albumin Injection in Pulmonary Malignancies: Feasibility Evaluation of Bronchial Artery 90Y Radioembolization.Radiology. 2025 Feb;314(2):e240331. doi: 10.1148/radiol.240331. Radiology. 2025. PMID: 39964268 Clinical Trial.
References
-
- Bray F, Ferlay J, Soerjomataram I, et al.: Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424, 2018 - PubMed
-
- Tyldesley S, Boyd C, Schulze K, et al.: Estimating the need for radiotherapy for lung cancer: an evidence-based, epidemiologic approach. Int J Radiat Oncol Biol Phys 49:973–85, 2001 - PubMed
-
- Bradley JD, Paulus R, Komaki R, et al.: Standard-dose versus high-dose conformal radiotherapy with concurrent and consolidation carboplatin plus paclitaxel with or without cetuximab for patients with stage IIIA or IIIB non-small-cell lung cancer (RTOG 0617): a randomised, two-by-two factorial phase 3 study. Lancet Oncol 16:187–99, 2015 - PMC - PubMed
-
- Senan S, Brade A, Wang LH, et al.: PROCLAIM: Randomized phase III trial of pemetrexed-cisplatin or etoposide-cisplatin plus thoracic radiation therapy followed by consolidation chemotherapy in locally advanced nonsquamous non-small-cell lung cancer. J Clin Oncol 34:953–62, 2016 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

