Cholinergic receptors on intestine cells of Ascaris suum and activation of nAChRs by levamisole
- PMID: 32470835
- PMCID: PMC7256660
- DOI: 10.1016/j.ijpddr.2020.04.002
Cholinergic receptors on intestine cells of Ascaris suum and activation of nAChRs by levamisole
Abstract
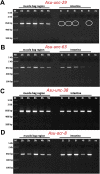
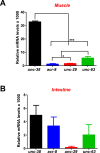
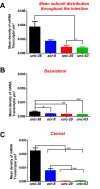
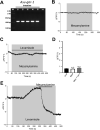
Cholinergic agonists, like levamisole, are a major class of anthelmintic drugs that are known to act selectively on nicotinic acetylcholine receptors (nAChRs) on the somatic muscle and nerves of nematode parasites to produce their contraction and spastic paralysis. Previous studies have suggested that in addition to the nAChRs found on muscle and nerves, there are nAChRs on non-excitable tissues of nematode parasites. We looked for evidence of nAChRs expression in the cells of the intestine of the large pig nematode, Ascaris suum, using RT-PCR and RNAscope in situ hybridization and detected mRNA of nAChR subunits in the cells. These subunits include components of the putative levamisole receptor in A. suum muscle: Asu-unc-38, Asu-unc-29, Asu-unc-63 and Asu-acr-8. Relative expression of these mRNAs in A. suum intestine was quantified by qPCR. We also looked for and found expression of G protein-linked acetylcholine receptors (Asu-gar-1). We used Fluo-3 AM to detect intracellular calcium changes in response to receptor activation by acetylcholine (as a non-selective agonist) and levamisole (as an L-type nAChR agonist) to look for evidence of functioning nAChRs in the intestine. We found that both acetylcholine and levamisole elicited increases in intracellular calcium but their signal profiles in isolated intestinal tissues were different, suggesting activation of different receptor sets. The levamisole responses were blocked by mecamylamine, a nicotinic receptor antagonist in A. suum, indicating the activation of intestinal nAChRs rather than G protein-linked acetylcholine receptors (GARs) by levamisole. The detection of nAChRs in cells of the intestine, in addition to those on muscles and nerves, reveals another site of action of the cholinergic anthelmintics and a site that may contribute to the synergistic interactions of cholinergic anthelmintics with other anthelmintics that affect the intestine (Cry5B).
Keywords: Calcium signaling; Intestine; Levamisole; Nicotinic acetylcholine receptors.
Copyright © 2020 The Authors. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare no conflict of interests.
Figures




Similar articles
-
Pharmacological profile of Ascaris suum ACR-16, a new homomeric nicotinic acetylcholine receptor widely distributed in Ascaris tissues.Br J Pharmacol. 2016 Aug;173(16):2463-77. doi: 10.1111/bph.13524. Epub 2016 Jul 18. Br J Pharmacol. 2016. PMID: 27238203 Free PMC article.
-
The nicotinic acetylcholine receptors of the parasitic nematode Ascaris suum: formation of two distinct drug targets by varying the relative expression levels of two subunits.PLoS Pathog. 2009 Jul;5(7):e1000517. doi: 10.1371/journal.ppat.1000517. Epub 2009 Jul 17. PLoS Pathog. 2009. PMID: 19609360 Free PMC article.
-
Monepantel is a non-competitive antagonist of nicotinic acetylcholine receptors from Ascaris suum and Oesophagostomum dentatum.Int J Parasitol Drugs Drug Resist. 2018 Apr;8(1):36-42. doi: 10.1016/j.ijpddr.2017.12.001. Epub 2017 Dec 16. Int J Parasitol Drugs Drug Resist. 2018. PMID: 29366967 Free PMC article.
-
Nicotinic acetylcholine receptors: a comparison of the nAChRs of Caenorhabditis elegans and parasitic nematodes.Parasitol Int. 2013 Dec;62(6):606-15. doi: 10.1016/j.parint.2013.03.004. Epub 2013 Mar 15. Parasitol Int. 2013. PMID: 23500392 Review.
-
Actions of neurotoxins (bungarotoxins, neosurugatoxin and lophotoxins) on insect and nematode nicotinic acetylcholine receptors.Toxicon. 1995 Apr;33(4):411-24. doi: 10.1016/0041-0101(94)00163-3. Toxicon. 1995. PMID: 7570627 Review.
Cited by
-
Diethylcarbamazine, TRP channels and Ca2+ signaling in cells of the Ascaris intestine.Sci Rep. 2022 Dec 9;12(1):21317. doi: 10.1038/s41598-022-25648-7. Sci Rep. 2022. PMID: 36494409 Free PMC article.
-
Dual SPE-HPLC-MS/MS Platform for Cross-Class Antiparasitic Surveillance: Simultaneous Quantification of Oxyclozanide and Levamisole Hydrochloride in Ovine Tissues with Applications to Withdrawal Period Optimization.Molecules. 2025 Mar 26;30(7):1473. doi: 10.3390/molecules30071473. Molecules. 2025. PMID: 40286083 Free PMC article.
-
Caenorhabditis elegans Neurotoxicity Testing: Novel Applications in the Adverse Outcome Pathway Framework.Front Toxicol. 2022 Mar 16;4:826488. doi: 10.3389/ftox.2022.826488. eCollection 2022. Front Toxicol. 2022. PMID: 35373186 Free PMC article.
-
The nematode (Ascaris suum) intestine is a location of synergistic anthelmintic effects of Cry5B and levamisole.PLoS Pathog. 2024 May 17;20(5):e1011835. doi: 10.1371/journal.ppat.1011835. eCollection 2024 May. PLoS Pathog. 2024. PMID: 38758969 Free PMC article.
-
Anthelmintic resistance and homeostatic plasticity (Brugia malayi).Sci Rep. 2021 Jul 14;11(1):14499. doi: 10.1038/s41598-021-93911-4. Sci Rep. 2021. PMID: 34262123 Free PMC article.
References
-
- Alexander P.E., De P. HIV-1 and intestinal helminth review update: updating a Cochrane Review and building the case for treatment and has the time come to test and treat? Parasite Immunol. 2009;31:283–286. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

