Murine cytomegalovirus dissemination but not reactivation in donor-positive/recipient-negative allogeneic kidney transplantation can be effectively prevented by transplant immune tolerance
- PMID: 32471635
- PMCID: PMC7311252
- DOI: 10.1016/j.kint.2020.01.034
Murine cytomegalovirus dissemination but not reactivation in donor-positive/recipient-negative allogeneic kidney transplantation can be effectively prevented by transplant immune tolerance
Abstract
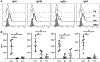
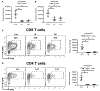
Cytomegalovirus (CMV) reactivation from latently infected donor organs post-transplantation and its dissemination cause significant comorbidities in transplant recipients. Transplant-induced inflammation combined with chronic immunosuppression has been thought to provoke CMV reactivation and dissemination, although sequential events in this process have not been studied. Here, we investigated this process in a high-risk donor CMV-positive to recipient CMV-negative allogeneic murine kidney transplantation model. Recipients were either treated with indefinite immunosuppression or tolerized in a donor-specific manner. Untreated recipients served as controls. Kidney allografts from both immunosuppressed and tolerized recipients showed minimal alloimmunity-mediated graft inflammation and normal function for up to day 60 post-transplantation. However, despite the absence of such inflammation in the immunosuppressed and tolerized groups, CMV reactivation in the donor positive kidney allograft was readily observed. Interestingly, subsequent CMV replication and dissemination to distant organs only occurred in immunosuppressed recipients in which CMV-specific CD8 T cells were functionally impaired; whereas in tolerized recipients, host anti-viral immunity was well-preserved and CMV dissemination was effectively prevented. Thus, our studies uncoupled CMV reactivation from its dissemination, and underscore the potential role of robust transplantation tolerance in preventing CMV diseases following allogeneic kidney transplantation.
Keywords: apoptotic cell therapy; cytomegalovirus reactivation; immunosuppression; kidney transplant; transplant immune tolerance.
Copyright © 2020 International Society of Nephrology. Published by Elsevier Inc. All rights reserved.
Figures

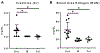
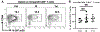


Similar articles
-
Acute murine cytomegalovirus disrupts established transplantation tolerance and causes recipient allo-sensitization.Am J Transplant. 2021 Feb;21(2):515-524. doi: 10.1111/ajt.16197. Epub 2020 Aug 17. Am J Transplant. 2021. PMID: 32659030 Free PMC article.
-
MCMV Dissemination from Latently-Infected Allografts Following Transplantation into Pre-Tolerized Recipients.Pathogens. 2020 Jul 26;9(8):607. doi: 10.3390/pathogens9080607. Pathogens. 2020. PMID: 32722544 Free PMC article.
-
A clinically relevant murine model unmasks a "two-hit" mechanism for reactivation and dissemination of cytomegalovirus after kidney transplant.Am J Transplant. 2019 Sep;19(9):2421-2433. doi: 10.1111/ajt.15376. Epub 2019 May 14. Am J Transplant. 2019. PMID: 30947382 Free PMC article.
-
A model for reactivation of CMV from latency.J Clin Virol. 2002 Aug;25 Suppl 2:S123-36. doi: 10.1016/s1386-6532(02)00088-4. J Clin Virol. 2002. PMID: 12361763 Review.
-
Potential Beneficial Effects of Cytomegalovirus Infection after Transplantation.Front Immunol. 2018 Mar 1;9:389. doi: 10.3389/fimmu.2018.00389. eCollection 2018. Front Immunol. 2018. PMID: 29545802 Free PMC article. Review.
Cited by
-
Can mouse kidney transplant models inform mechanisms of injury and acceptance in clinical kidney transplantation?Am J Transplant. 2025 Jul;25(7):1391-1398. doi: 10.1016/j.ajt.2025.04.001. Epub 2025 Apr 9. Am J Transplant. 2025. PMID: 40209906 Free PMC article. Review.
-
Acute murine cytomegalovirus disrupts established transplantation tolerance and causes recipient allo-sensitization.Am J Transplant. 2021 Feb;21(2):515-524. doi: 10.1111/ajt.16197. Epub 2020 Aug 17. Am J Transplant. 2021. PMID: 32659030 Free PMC article.
-
Diverse Routes of Allograft Tolerance Disruption by Memory T Cells.Front Immunol. 2020 Oct 8;11:580483. doi: 10.3389/fimmu.2020.580483. eCollection 2020. Front Immunol. 2020. PMID: 33117387 Free PMC article. Review.
-
MCMV Dissemination from Latently-Infected Allografts Following Transplantation into Pre-Tolerized Recipients.Pathogens. 2020 Jul 26;9(8):607. doi: 10.3390/pathogens9080607. Pathogens. 2020. PMID: 32722544 Free PMC article.
-
Single cell transcriptomics of mouse kidney transplants reveals a myeloid cell pathway for transplant rejection.JCI Insight. 2020 Oct 15;5(20):e141321. doi: 10.1172/jci.insight.141321. JCI Insight. 2020. PMID: 32970632 Free PMC article.
References
-
- Fishman JA, Emery V, Freeman R, et al. Cytomegalovirus in transplantation - challenging the status quo. Clinical transplantation 2007; 21: 149–158. - PubMed
-
- Sagedal S, Nordal KP, Hartmann A, et al. A prospective study of the natural course of cytomegalovirus infection and disease in renal allograft recipients. Transplantation 2000; 70: 1166–1174. - PubMed
-
- Sinclair J, Sissons P. Latency and reactivation of human cytomegalovirus. The Journal of general virology 2006; 87: 1763–1779. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

