Proteomic Profiling of Plasma and Brain Tissue from Alzheimer's Disease Patients Reveals Candidate Network of Plasma Biomarkers
- PMID: 32474469
- PMCID: PMC7457324
- DOI: 10.3233/JAD-200110
Proteomic Profiling of Plasma and Brain Tissue from Alzheimer's Disease Patients Reveals Candidate Network of Plasma Biomarkers
Abstract
Background: Alzheimer's disease (AD) is the most prevalent form of dementia with two pathological hallmarks of tau-containing neurofibrillary tangles and amyloid-β protein (Aβ)-containing neuritic plaques. Although Aβ and tau have been explored as potential biomarkers, levels of these pathological proteins in blood fail to distinguish AD from healthy control subjects.
Objective: We aim to discover potential plasma proteins associated with AD pathology by performing tandem mass tag (TMT)-based quantitative proteomic analysis of proteins from peripheral and central nervous system compartments.
Methods: We performed comparative proteomic analyses of plasma collected from AD patients and cognitively normal subjects. In addition, proteomic profiles from the inferior frontal cortex, superior frontal cortex, and cerebellum of postmortem brain tissue from five AD patients and five non-AD controls were compared with plasma proteomic profiles to search for common biomarkers. Liquid chromatography-mass spectrometry was used to analyze plasma and brain tissue labeled with isobaric TMT for relative protein quantification.
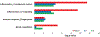
Results: Our results showed that the proteins in complement coagulation cascade and interleukin-6 signaling were significantly altered in both plasma and brains of AD patients.
Conclusion: Our results demonstrate the relevance in immune responses between the peripheral and central nervous systems. Those differentially regulated plasma proteins are explored as candidate biomarker profiles that illustrate chronic neuroinflammation in brains of AD patients.
Keywords: Alzheimer’s disease; biomarker; mass spectrometry; plasma; quantitative proteomics; tandem mass tag (TMT).
Figures




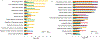
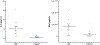
References
-
- Selkoe DJ (2004) Alzheimer disease: Mechanistic understanding predicts novel therapies. Ann Intern Med 140, 627–638. - PubMed
-
- Xia W (2001) Amyloid metabolism and secretases in Alzheimer’s disease. Curr Neurol Neurosci Rep 1, 422–427. - PubMed
-
- Heppner FL, Ransohoff RM, Becher B (2015) Immune attack: The role of inflammation in Alzheimer disease. Nat Rev Neurosci 16, 358–372. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

