Novel Role for Tranilast in Regulating NLRP3 Ubiquitination, Vascular Inflammation, and Atherosclerosis
- PMID: 32476536
- PMCID: PMC7429049
- DOI: 10.1161/JAHA.119.015513
Novel Role for Tranilast in Regulating NLRP3 Ubiquitination, Vascular Inflammation, and Atherosclerosis
Abstract
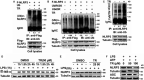
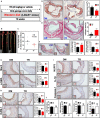
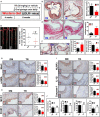
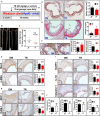
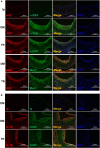
Background Aberrant activation of the NLRP3 (nucleotide-binding oligomerization domain, leucine-rich repeat-containing receptor family pyrin domain-containing 3) inflammasome is thought to play a causative role in atherosclerosis. NLRP3 is kept in an inactive ubiquitinated state to avoid unwanted NLRP3 inflammasome activation. This study aimed to test the hypothesis that pharmacologic manipulating of NLRP3 ubiquitination blunts the assembly and activation of the NLRP3 inflammasome and protects against vascular inflammation and atherosclerosis. Since genetic studies yielded mixed results about the role for this inflammasome in atherosclerosis in low-density lipoprotein receptor- or apolipoprotein E-deficient mice, this study attempted to clarify the discrepancy with the pharmacologic approach using both models. Methods and Results We provided the first evidence demonstrating that tranilast facilitates NLRP3 ubiquitination. We showed that tranilast restricted NLRP3 oligomerization and inhibited NLRP3 inflammasome assembly. Tranilast markedly suppressed NLRP3 inflammasome activation in low-density lipoprotein receptor- and apolipoprotein E-deficient macrophages. Through reconstitution of the NLRP3 inflammasome in human embryonic kidney 293T cells, we found that tranilast directly limited NLRP3 inflammasome activation. By adopting different regimens for tranilast treatment of low-density lipoprotein receptor- and apolipoprotein E-deficient mice, we demonstrated that tranilast blunted the initiation and progression of atherosclerosis. Mice receiving tranilast displayed a significant reduction in atherosclerotic lesion size, concomitant with a pronounced decline in macrophage content and expression of inflammatory molecules in the plaques compared with the control group. Moreover, tranilast treatment of mice substantially hindered the expression and activation of the NLRP3 inflammasome in the atherosclerotic lesions. Conclusions Tranilast potently enhances NLRP3 ubiquitination, blunts the assembly and activation of the NLRP3 inflammasome, and ameliorates vascular inflammation and atherosclerosis in both low-density lipoprotein receptor- and apolipoprotein E-deficient mice.
Keywords: NLRP3 ubiquitination; atherosclerosis; inflammasome; tranilast; vascular inflammation.
Figures



References
-
- Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377:1119–1131. - PubMed
-
- Voronov E, Apte RN. Targeting the tumor microenvironment by intervention in interleukin‐1 biology. Curr Pharm Des. 2017;23:4893–4905. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

