Herpes Simplex Virus 1 UL2 Inhibits the TNF-α-Mediated NF-κB Activity by Interacting With p65/p50
- PMID: 32477319
- PMCID: PMC7237644
- DOI: 10.3389/fimmu.2020.00549
Herpes Simplex Virus 1 UL2 Inhibits the TNF-α-Mediated NF-κB Activity by Interacting With p65/p50
Abstract
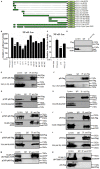
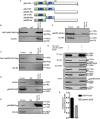
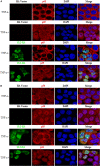
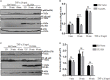
Herpes simplex virus 1 (HSV-1) is a large double-stranded DNA virus that encodes at least 80 viral proteins, many of which are involved in the virus-host interaction and are beneficial to the viral survival and reproduction. However, the biological functions of some HSV-1-encoded proteins are not fully understood. Nuclear factor κB (NF-κB) activation is the major antiviral innate response, which can be triggered by various signals induced by cellular receptors from different pathways. Here, we demonstrated that HSV-1 UL2 protein could antagonize the tumor necrosis factor α (TNF-α)-mediated NF-κB activation. Co-immunoprecipitation assays showed that UL2 could interact with the NF-κB subunits p65 and p50, which also revealed the region of amino acids 9 to 17 of UL2 could suppress the NF-κB activation and interact with p65 and p50, and UL2 bound to the immunoglobulin-like plexin transcription factor functional domain of p65. However, UL2 did not affect the formation of p65/p50 dimerization and their nuclear localizations. Yet, UL2 was demonstrated to inhibit the NF-κB activity by attenuating TNF-α-induced p65 phosphorylation at Ser536 and therefore decreasing the expression of downstream inflammatory chemokine interleukin 8. Taken together, the attenuation of NF-κB activation by UL2 may contribute to the escape of host's antiviral innate immunity for HSV-1 during its infection.
Keywords: HSV-1; IL-8; NF-κB; UL2; innate immunity.
Copyright © 2020 Cai, Liao, Zou, Xu, Wang, Li, Li, Ou, Deng, Guo, Peng and Li.
Figures





Similar articles
-
Herpes simplex virus 1 DNA polymerase processivity factor UL42 inhibits TNF-α-induced NF-κB activation by interacting with p65/RelA and p50/NF-κB1.Med Microbiol Immunol. 2013 Aug;202(4):313-25. doi: 10.1007/s00430-013-0295-0. Epub 2013 May 1. Med Microbiol Immunol. 2013. PMID: 23636254
-
Herpes simplex virus 1 E3 ubiquitin ligase ICP0 protein inhibits tumor necrosis factor alpha-induced NF-κB activation by interacting with p65/RelA and p50/NF-κB1.J Virol. 2013 Dec;87(23):12935-48. doi: 10.1128/JVI.01952-13. Epub 2013 Sep 25. J Virol. 2013. PMID: 24067962 Free PMC article.
-
Herpes Simplex Virus 1 UL24 Abrogates the DNA Sensing Signal Pathway by Inhibiting NF-κB Activation.J Virol. 2017 Mar 13;91(7):e00025-17. doi: 10.1128/JVI.00025-17. Print 2017 Apr 1. J Virol. 2017. PMID: 28100608 Free PMC article.
-
Suppression of NF-κB Activity: A Viral Immune Evasion Mechanism.Viruses. 2018 Aug 4;10(8):409. doi: 10.3390/v10080409. Viruses. 2018. PMID: 30081579 Free PMC article. Review.
-
The Race between Host Antiviral Innate Immunity and the Immune Evasion Strategies of Herpes Simplex Virus 1.Microbiol Mol Biol Rev. 2020 Sep 30;84(4):e00099-20. doi: 10.1128/MMBR.00099-20. Print 2020 Nov 18. Microbiol Mol Biol Rev. 2020. PMID: 32998978 Free PMC article. Review.
Cited by
-
The Molecular Mechanism of Herpes Simplex Virus 1 UL31 in Antagonizing the Activity of IFN-β.Microbiol Spectr. 2022 Feb 23;10(1):e0188321. doi: 10.1128/spectrum.01883-21. Epub 2022 Feb 23. Microbiol Spectr. 2022. PMID: 35196784 Free PMC article.
-
Ikzf2 Regulates the Development of ICOS+ Th Cells to Mediate Immune Response in the Spleen of S. japonicum-Infected C57BL/6 Mice.Front Immunol. 2021 Aug 12;12:687919. doi: 10.3389/fimmu.2021.687919. eCollection 2021. Front Immunol. 2021. PMID: 34475870 Free PMC article.
-
Herpes simplex virus type 2 inhibits TNF-α-induced NF-κB activation through viral protein ICP22-mediated interaction with p65.Front Immunol. 2022 Sep 23;13:983502. doi: 10.3389/fimmu.2022.983502. eCollection 2022. Front Immunol. 2022. PMID: 36211339 Free PMC article.
-
Curcumin can improve spinal cord injury by inhibiting DNA methylation.Mol Cell Biochem. 2024 Feb;479(2):351-362. doi: 10.1007/s11010-023-04731-1. Epub 2023 Apr 19. Mol Cell Biochem. 2024. PMID: 37076656
-
Antiviral Potential of Naphthoquinones Derivatives Encapsulated within Liposomes.Molecules. 2021 Oct 25;26(21):6440. doi: 10.3390/molecules26216440. Molecules. 2021. PMID: 34770849 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

