Structural Characteristics, Binding Partners and Related Diseases of the Calponin Homology (CH) Domain
- PMID: 32478077
- PMCID: PMC7240100
- DOI: 10.3389/fcell.2020.00342
Structural Characteristics, Binding Partners and Related Diseases of the Calponin Homology (CH) Domain
Abstract
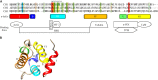
The calponin homology (CH) domain is one of the most common modules in various actin-binding proteins and is characterized by an α-helical fold. The CH domain plays important regulatory roles in both cytoskeletal dynamics and signaling. The CH domain is required for stability and organization of the actin cytoskeleton, calcium mobilization and activation of downstream pathways. The CH domain has recently garnered increased attention due to its importance in the onset of different diseases, such as cancers and asthma. However, many roles of the CH domain in various protein functions and corresponding diseases are still unclear. Here, we review current knowledge about the structural features, interactome and related diseases of the CH domain.
Keywords: CH-domain-containing proteins; actin cytoskeleton; calmodulin; cancer; transgelin-2; tropomyosin; tubulin; α-helix.
Copyright © 2020 Yin, Schnoor and Jun.
Figures
Similar articles
-
Solution structure of the calponin CH domain and fitting to the 3D-helical reconstruction of F-actin:calponin.Structure. 2002 Feb;10(2):249-58. doi: 10.1016/s0969-2126(02)00703-7. Structure. 2002. PMID: 11839310
-
The CH-domain of calponin does not determine the modes of calponin binding to F-actin.J Mol Biol. 2006 Jun 2;359(2):478-85. doi: 10.1016/j.jmb.2006.03.044. Epub 2006 Apr 3. J Mol Biol. 2006. PMID: 16626733
-
Extracellular regulated kinase (ERK) interaction with actin and the calponin homology (CH) domain of actin-binding proteins.Biochem J. 1999 Nov 15;344 Pt 1(Pt 1):117-23. Biochem J. 1999. PMID: 10548541 Free PMC article.
-
Novel structural insights into F-actin-binding and novel functions of calponin homology domains.Curr Opin Struct Biol. 2008 Dec;18(6):702-8. doi: 10.1016/j.sbi.2008.10.003. Epub 2008 Nov 13. Curr Opin Struct Biol. 2008. PMID: 18952167 Review.
-
Some properties of caldesmon and calponin and the participation of these proteins in regulation of smooth muscle contraction and cytoskeleton formation.Biochemistry (Mosc). 2001 Oct;66(10):1112-21. doi: 10.1023/a:1012480829618. Biochemistry (Mosc). 2001. PMID: 11736632 Review.
Cited by
-
MFN2 deficiency affects calcium homeostasis in lung adenocarcinoma cells via downregulation of UCP4.FEBS Open Bio. 2023 Jun;13(6):1107-1124. doi: 10.1002/2211-5463.13591. Epub 2023 Mar 14. FEBS Open Bio. 2023. PMID: 36877954 Free PMC article.
-
Transgelins: Cytoskeletal Associated Proteins Implicated in the Metastasis of Colorectal Cancer.Front Cell Dev Biol. 2020 Oct 7;8:573859. doi: 10.3389/fcell.2020.573859. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 33117801 Free PMC article. Review.
-
Re-evaluating the actin-dependence of spectraplakin functions during axon growth and maintenance.Dev Neurobiol. 2022 May;82(4):288-307. doi: 10.1002/dneu.22873. Epub 2022 Apr 22. Dev Neurobiol. 2022. PMID: 35333003 Free PMC article.
-
Structure and Emerging Functions of LRCH Proteins in Leukocyte Biology.Front Cell Dev Biol. 2020 Sep 22;8:584134. doi: 10.3389/fcell.2020.584134. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 33072765 Free PMC article. Review.
-
Expanding the repertoire of human tandem repeat RNA-binding proteins.PLoS One. 2023 Sep 20;18(9):e0290890. doi: 10.1371/journal.pone.0290890. eCollection 2023. PLoS One. 2023. PMID: 37729217 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources

