Role of Blood Oxygen Saturation During Post-Natal Human Cardiomyocyte Cell Cycle Activities
- PMID: 32478207
- PMCID: PMC7251192
- DOI: 10.1016/j.jacbts.2020.02.008
Role of Blood Oxygen Saturation During Post-Natal Human Cardiomyocyte Cell Cycle Activities
Abstract
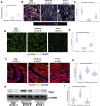
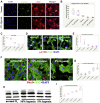
Blood oxygen saturation (SaO2) is one of the most important environmental factors in clinical heart protection. This study used human heart samples and human induced pluripotent stem cell-cardiomyocytes (iPSC-CMs) to assess how SaO2 affects human CM cell cycle activities. The results showed that there were significantly more cell cycle markers in the moderate hypoxia group (SaO2: 75% to 85%) than in the other 2 groups (SaO2 <75% or >85%). In iPSC-CMs 15% and 10% oxygen (O2) treatment increased cell cycle markers, whereas 5% and rapid change of O2 decreased the markers. Moderate hypoxia is beneficial to the cell cycle activities of post-natal human CMs.
Keywords: CHD, congenital heart disease; CM, cardiomyocytes; IF, immunofluorescence; LV, lentivirus; O2, oxygen; SaO2, blood oxygen saturation; TOF, tetralogy of Fallot; YAP1, yes-associated protein 1; blood oxygen saturation; cardiomyocyte; congenital heart disease; iPSC, induced pluripotent stem cell; pATM, phosphorylated ataxia telangiectasia mutated; pHH3, phospho-histone H3; pediatric patients; proliferation; qPCR, quantitative polymerase chain reaction; sh, short hairpin.
© 2020 The Authors.
Figures





Comment in
-
Hypoxia Induces Cardiomyocyte Proliferation in Humans.JACC Basic Transl Sci. 2020 May 25;5(5):461-462. doi: 10.1016/j.jacbts.2020.04.008. eCollection 2020 May. JACC Basic Transl Sci. 2020. PMID: 32479579 Free PMC article. No abstract available.
References
-
- Tennant P.W., Pearce M.S., Bythell M., Rankin J. 20-year survival of children born with congenital anomalies: a population-based study. Lancet. 2010;375:649–656. - PubMed
-
- Hoffman J.I., Kaplan S. The incidence of congenital heart disease. J Am Coll Cardiol. 2002;39:1890–1900. - PubMed
-
- Bolger A.P., Coats A.J., Gatzoulis M.A. Congenital heart disease: the original heart failure syndrome. Eur Heart J. 2003;24:970–976. - PubMed
-
- Cerillo A.G., Storti S., Kallushi E. The low triiodothyronine syndrome: a strong predictor of low cardiac output and death in patients undergoing coronary artery bypass grafting. Ann Thorac Surg. 2014;97:2089–2095. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

