Are Supplements Safe? Effects of Gallic and Ferulic Acids on In Vitro Cell Models
- PMID: 32485864
- PMCID: PMC7352663
- DOI: 10.3390/nu12061591
Are Supplements Safe? Effects of Gallic and Ferulic Acids on In Vitro Cell Models
Abstract
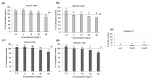
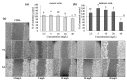
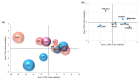
Polyphenols display health-promoting properties linked to their biological activities. They are initially absorbed in the small intestine, then they are largely metabolized in the colon, whereupon they are able to exert systemic effects. The health-promoting properties of polyphenols have led to the development of food supplements, which are also largely consumed by healthy people, even if data on their safety are still yet lacking. In the present paper, the content of gallic acid and ferulic acid was analyzed in two supplements, and shown to be higher than the relative contents found in fruit and flour. To evaluate the effects of these phenolic compounds on epithelial intestinal tissue, gallic and ferulic acids were added to a new in vitro model of the intestinal wall at different concentrations. The effects on viability, proliferation and migration of these compounds were respectively tested on three different cell lines (Caco2, L929 and U937), as well as on a tridimensional intestinal model, composed of a mucosal layer and a submucosa with fibroblasts and monocytes. Results indicated that gallic and ferulic acids can exert toxic effects on in vitro cell models at high concentrations, suggesting that an excessive and uncontrolled consumption of polyphenols may induce negative effects on the intestinal wall.
Keywords: ferulic acid; gallic acid; intestinal wall models; supplements.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

