Lipid Metabolism in Development and Progression of Hepatocellular Carcinoma
- PMID: 32486341
- PMCID: PMC7352397
- DOI: 10.3390/cancers12061419
Lipid Metabolism in Development and Progression of Hepatocellular Carcinoma
Abstract
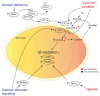
: Metabolic reprogramming is critically involved in the development and progression of cancer. In particular, lipid metabolism has been investigated as a source of energy, micro-environmental adaptation, and cell signalling in neoplastic cells. However, the specific role of lipid metabolism dysregulation in hepatocellular carcinoma (HCC) has not been widely described yet. Alterations in fatty acid synthesis, β-oxidation, and cellular lipidic composition contribute to initiation and progression of HCC. The aim of this review is to elucidate the mechanisms by which lipid metabolism is involved in hepatocarcinogenesis and tumour adaptation to different conditions, focusing on the transcriptional aberrations with new insights in lipidomics and lipid zonation. This will help detect new putative therapeutic approaches in the second most frequent cause of cancer-related death.
Keywords: fatty acid β-oxidation; hepatocellular carcinoma; lipid metabolism; lipidomics; non-alcoholic fatty liver disease; tumour progression.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Gluer A.M., Cocco N., Laurence J.M., Johnston E.S., Hollands M.J., Pleass H.C., Richardson A.J., Lam V.W. Systematic review of actual 10-year survival following resection for hepatocellular carcinoma. HPB Off. J. Int. Hepato Pancreato Biliary Assoc. 2012;14:285–290. doi: 10.1111/j.1477-2574.2012.00446.x. - DOI - PMC - PubMed
-
- Shiina S., Tateishi R., Arano T., Uchino K., Enooku K., Nakagawa H., Asaoka Y., Sato T., Masuzaki R., Kondo Y., et al. Radiofrequency ablation for hepatocellular carcinoma: 10-year outcome and prognostic factors. Am. J. Gastroenterol. 2012;107:569–577. doi: 10.1038/ajg.2011.425. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

