Galectins and Ovarian Cancer
- PMID: 32486344
- PMCID: PMC7352943
- DOI: 10.3390/cancers12061421
Galectins and Ovarian Cancer
Abstract
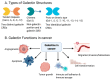
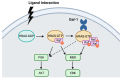
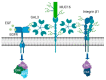
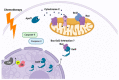
Ovarian cancer is known for its aggressive pathological features, including the capacity to undergo epithelial to mesenchymal transition, promoting angiogenesis, metastatic potential, chemoresistance, inhibiting apoptosis, immunosuppression and promoting stem-like features. Galectins, a family of glycan-binding proteins defined by a conserved carbohydrate recognition domain, can modulate many of these processes, enabling them to contribute to the pathology of ovarian cancer. Our goal herein was to review specific galectin members identified in the context of ovarian cancer, with emphasis on their association with clinical and pathological features, implied functions, diagnostic or prognostic potential and strategies being developed to disrupt their negative actions.
Keywords: chemoresistance; galectins; immune suppression; invasion; metastasis; ovarian cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Levi G., I Teichberg V. Isolation and physicochemical characterization of electrolectin, a beta-D-galactoside binding lectin from the electric organ of Electrophorus electricus. J. Boil. Chem. 1981;256:5735–5740. - PubMed
-
- Sakthivel D., Littler D., Shahine A., Troy S., Johnson M., Rossjohn J., Piedrafita D., Beddoe T. Cloning, expression, purification and crystallographic studies of galectin-11 from domestic sheep (Ovis aries) Acta Crystallogr. Sect. F Struct. Boil. Commun. 2015;71:993–997. doi: 10.1107/S2053230X15010195. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

