Functional connectivity of EEG is subject-specific, associated with phenotype, and different from fMRI
- PMID: 32492509
- PMCID: PMC7457369
- DOI: 10.1016/j.neuroimage.2020.117001
Functional connectivity of EEG is subject-specific, associated with phenotype, and different from fMRI
Abstract
A variety of psychiatric, behavioral and cognitive phenotypes have been linked to brain ''functional connectivity'' -- the pattern of correlation observed between different brain regions. Most commonly assessed using functional magnetic resonance imaging (fMRI), here, we investigate the connectivity-phenotype associations with functional connectivity measured with electroencephalography (EEG), using phase-coupling. We analyzed data from the publicly available Healthy Brain Network Biobank. This database compiles a growing sample of children and adolescents, currently encompassing 1657 individuals. Among a variety of assessment instruments we focus on ten phenotypic and additional demographic measures that capture most of the variance in this sample. The largest effect sizes are found for age and sex for both fMRI and EEG. We replicate previous findings of an association of Intelligence Quotient (IQ) and Attention Deficit Hyperactivity Disorder (ADHD) with the pattern of fMRI functional connectivity. We also find an association with socioeconomic status, anxiety and the Child Behavior Checklist Score. For EEG we find a significant connectivity-phenotype relationship with IQ. The actual spatial patterns of functional connectivity are quite different between fMRI and source-space EEG. However, within EEG we observe clusters of functional connectivity that are consistent across frequency bands. Additionally we analyzed reproducibility of functional connectivity. We compare connectivity obtained with different tasks, including resting state, a video and a visual flicker task. For both EEG and fMRI the variation between tasks was smaller than the variability observed between subjects. We also found an increase of reliability with increasing frequency of the EEG, and increased sampling duration. We conclude that, while the patterns of functional connectivity are distinct between fMRI and phase-coupling of EEG, they are nonetheless similar in their robustness to the task, and similar in that idiosyncratic patterns of connectivity predict individual phenotypes.
Keywords: Brain–behavior relationships; Electroencephalography (EEG); Functional connectivity; Functional magnetic resonance imaging (fMRI); Imaginary coherence; Reliability.
Copyright © 2020 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare no conflict of interest.
Figures



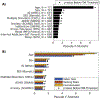


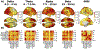
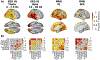
References
-
- Alexander LM, Escalera J, Ai L, Andreotti C, Febre K, Mangone A, Vega-Potler N, Langer N, Alexander A, Kovacs M, Litke S, O’Hagan B, Andersen J, Bronstein B, Bui A, Bushey M, Butler H, Castagna V, Camacho N, Chan E, Citera D, Clucas J, Cohen S, Dufek S, Eaves M, Fradera B, Gardner J, Grant-Villegas N, Green G, Gregory C, Hart E, Harris S, Horton M, Kahn D, Kabotyanski K, Karmel B, Kelly SP, Kleinman K, Koo B, Kramer E, Lennon E, Lord C, Mantello G, Margolis A, Merikangas KR, Milham J, Minniti G, Neuhaus R, Levine A, Osman Y, Parra LC, Pugh KR, Racanello A, Restrepo A, Saltzman T, Septimus B, Tobe R, Waltz R, Williams A, Yeo A, Castellanos FX, Klein A, Paus T, Leventhal BL, Craddock RC, Koplewicz HS, Milham MP, 2017. An open resource for transdiagnostic research in pediatric mental health and learning disorders. Sci. Data 4, 170181 10.1038/sdata.2017.181. - DOI - PMC - PubMed
-
- Benjamini Y, Hochberg Y, 1995. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B Methodol 57, 289–300. 10.1111/j.2517-6161.1995.tb02031.x. - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

