An evidence mapping and analysis of registered COVID-19 clinical trials in China
- PMID: 32493331
- PMCID: PMC7268588
- DOI: 10.1186/s12916-020-01612-y
An evidence mapping and analysis of registered COVID-19 clinical trials in China
Abstract
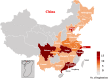
Background: This article aims to summarize the key characteristics of registered trials of 2019 novel coronavirus (COVID-19), in terms of their spatial and temporal distributions, types of design and interventions, and patient characteristics among others.
Methods: A comprehensive search of the registered COVID-19 trials has been performed on platforms including ClinicalTrials.gov, WHO International Clinical Trials Registry Platform (WHO ICTRP), Chinese Clinical Trials Registry (CHiCTR), Australian Clinical Trials Registry, Britain's National Research Register (BNRR), Current Control Trials (CCT), and Glaxo Smith Kline Register. Trials registered at the first 8 weeks of the COVID-19 outbreak are included, without language restrictions. For each study, the registration information, study design, and administrator information are collected and summarized.
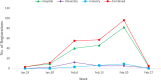
Results: A total of 220 registered trials were evaluated as of February 27, 2020. Hospital-initiated trials were the majority and account for 80% of the sample. Among the trials, pilot studies and phase 4 trials are more common and represent 35% and 19.1% of the sample, respectively. The median sample size of the registered trials is 100, with interquartile range 60-240. Further, 45.9% of the trials mentioned information on a data monitoring committee. 54.5% of the trials did not specify the disease severity among patients they intend to recruit. Four types of interventions are most common in the experimental groups across the registered studies: antiviral drugs, Traditional Chinese Medicine (TCM), biological agents, and hormone drugs. Among them, the TCM and biological agents are frequently used in pilot study and correspond to a variety of primary endpoints. In contrast, trials with antiviral drugs have more targeted primary outcomes such as "COVID-19 nucleic acid test" and "28-day mortality."
Conclusions: We provide an evidence mapping and analysis of registered COVID-19 clinical trials in China. In particular, it is critical for ongoing and future studies to refine their research hypothesis and better identify their intervention therapies and the corresponding primary outcomes. It is also imperative for multiple public health divisions and research institutions to work together for integrative clinical data capture and sharing, with a common objective of improving future studies that evaluate COVID-19 interventions.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- WHO. WHO Director-General’s remarks at the media briefing on 2019-nCoV on 11 February 2020. https://www.who.int/dg/speeches/detail/who-director-general-s-remarks-at... (Accessed 22 Feb 2020).
-
- People's Daily. The real-time dynamic monitoring data of 2019nCoV1 in China. http://health.people.com.cn/GB/26466/431463/431576/index.html (Accessed 22 Feb 2020).
Publication types
MeSH terms
Substances
Grants and funding
- UL1 TR001863/TR/NCATS NIH HHS/United States
- 2019QNPY02/Youth Scientific Research Training Project of GZUCM/International
- 2017TQ04R627/Young Top Talent Project of Scientific and Technological Innovation in Special Support Plan for Training High-level Talents in Guangdong/International
- 81873375/National Natural Science Foundation of China/International
LinkOut - more resources
Full Text Sources
Medical

