Evaluation of a novel antigen-based rapid detection test for the diagnosis of SARS-CoV-2 in respiratory samples
- PMID: 32497809
- PMCID: PMC7263236
- DOI: 10.1016/j.ijid.2020.05.098
Evaluation of a novel antigen-based rapid detection test for the diagnosis of SARS-CoV-2 in respiratory samples
Abstract
Objectives: In the context of the coronavirus disease 2019 (COVID-19) pandemic, the development and validation of rapid and easy-to-perform diagnostic methods are of high priority. This study was performed to evaluate a novel rapid antigen detection test (RDT) for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in respiratory samples.
Methods: The fluorescence immunochromatographic SARS-CoV-2 antigen test (Bioeasy Biotechnology Co., Shenzhen, China) was evaluated using universal transport medium with nasopharyngeal (NP) and oropharyngeal (OP) swabs from suspected COVID-19 cases. Diagnostic accuracy was determined in comparison to SARS-CoV-2 real-time (RT)-PCR.
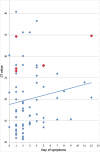
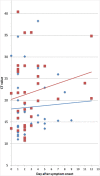
Results: A total of 127 samples were included; 82 were RT-PCR-positive. The median patient age was 38 years, 53.5% were male, and 93.7% were from the first week after symptom onset. Overall sensitivity and specificity were 93.9% (95% confidence interval 86.5-97.4%) and 100% (95% confidence interval 92.1-100%), respectively, with a diagnostic accuracy of 96.1% and Kappa coefficient of 0.9. Sensitivity was significantly higher in samples with high viral loads.
Conclusions: The RDT evaluated in this study showed a high sensitivity and specificity in samples mainly obtained during the first week of symptoms and with high viral loads, despite the use of a non-validated sample material. The assay has the potential to become an important tool for early diagnosis of SARS-CoV-2, particularly in situations with limited access to molecular methods.
Keywords: Antigen; COVID-19; Coronavirus; Diagnosis; Rapid diagnostic test; SARS-CoV-2.
Copyright © 2020 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures
References
-
- CDC (Centers for Disease Control and Prevention), Respiratory Viruses Branch, Division of Viral Diseases. Real-Time RT-PCR Panel for Detection 2019-Novel Coronavirus. Instructions for Use. 2020. https://www.cdc.gov/coronavirus/2019-ncov/lab/index.html?CDC_AA_refVal=h..., 2020 (accessed 15 May 2020).
-
- CLSI . Clinical and Laboratory Standards Institute; Wayne, PA: 2008. User Protocol for Evaluation of Qualitative Test Performance; Approved Guideline-Second Edition. CLSI document EP12-A2.
-
- Diao B., Wen K., Chen J., Liu Y., Yuan Z., Han C. 2020. Diagnosis of acute respiratory syndrome coronavirus 2 infection by detection of nucleocapsid protein.https://www.medrxiv.org/content/10.1101/2020.03.07.20032524v2 Preprint at. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

