Nano-Polyplexes Mediated Transfection of Runx2-shRNA Mitigates the Osteodifferentiation of Human Valvular Interstitial Cells
- PMID: 32498305
- PMCID: PMC7355966
- DOI: 10.3390/pharmaceutics12060507
Nano-Polyplexes Mediated Transfection of Runx2-shRNA Mitigates the Osteodifferentiation of Human Valvular Interstitial Cells
Abstract
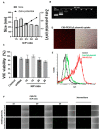
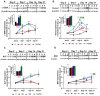
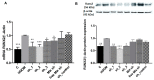
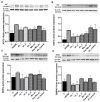
Calcific aortic valve disease (CAVD) is a progressive disorder that increases in prevalence with age. An important role in aortic valve calcification is played by valvular interstitial cells (VIC), that with age or in pathological conditions acquire an osteoblast-like phenotype that advances the disease. Therefore, pharmacological interventions aiming to stop or reverse the osteoblastic transition of VIC may represent a therapeutic option for CAVD. In this study, we aimed at developing a nanotherapeutic strategy able to prevent the phenotypic switch of human aortic VIC into osteoblast-like cells. We hypothesize that nanocarriers designed for silencing the Runt-related transcription factor 2 (Runx2) will stop the progress or reverse the osteodifferentiation of human VIC, induced by high glucose concentrations and pro-osteogenic factors. We report here the potential of fullerene (C60)-polyethyleneimine (PEI)/short hairpin (sh)RNA-Runx2 nano-polyplexes to efficiently down-regulate Runx2 mRNA and protein expression leading subsequently to a significant reduction in the expression of osteogenic proteins (i.e. ALP, BSP, OSP and BMP4) in osteoblast-committed VIC. The data suggest that the silencing of Runx2 could represent a novel strategy to impede the osteoblastic phenotypic shift of VIC and the ensuing progress of CAVD.
Keywords: Runx2; calcific aortic valve disease; nanocarriers; osteodifferentiation.; shRNA; valvular interstitial cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
VCAM-1 Targeted Lipopolyplexes as Vehicles for Efficient Delivery of shRNA-Runx2 to Osteoblast-Differentiated Valvular Interstitial Cells; Implications in Calcific Valve Disease Treatment.Int J Mol Sci. 2022 Mar 30;23(7):3824. doi: 10.3390/ijms23073824. Int J Mol Sci. 2022. PMID: 35409184 Free PMC article.
-
Nanocarriers of shRNA-Runx2 directed to collagen IV as a nanotherapeutic system to target calcific aortic valve disease.Mater Today Bio. 2023 Mar 28;20:100620. doi: 10.1016/j.mtbio.2023.100620. eCollection 2023 Jun. Mater Today Bio. 2023. PMID: 37063777 Free PMC article.
-
Hydrogen sulfide inhibits aortic valve calcification in heart via regulating RUNX2 by NF-κB, a link between inflammation and mineralization.J Adv Res. 2020 Jul 21;27:165-176. doi: 10.1016/j.jare.2020.07.005. eCollection 2021 Jan. J Adv Res. 2020. PMID: 33318875 Free PMC article. Review.
-
Simulation of early calcific aortic valve disease in a 3D platform: A role for myofibroblast differentiation.J Mol Cell Cardiol. 2016 May;94:13-20. doi: 10.1016/j.yjmcc.2016.03.004. Epub 2016 Mar 17. J Mol Cell Cardiol. 2016. PMID: 26996755 Free PMC article.
-
Toll-Like Receptors, Inflammation, and Calcific Aortic Valve Disease.Front Physiol. 2018 Mar 12;9:201. doi: 10.3389/fphys.2018.00201. eCollection 2018. Front Physiol. 2018. PMID: 29593562 Free PMC article. Review.
Cited by
-
Bioinformatics-Based Identification of CircRNA-MicroRNA-mRNA Network for Calcific Aortic Valve Disease.Genet Res (Camb). 2023 May 17;2023:8194338. doi: 10.1155/2023/8194338. eCollection 2023. Genet Res (Camb). 2023. PMID: 37234568 Free PMC article.
-
Emerging Gene Therapy Based on Nanocarriers: A Promising Therapeutic Alternative for Cardiovascular Diseases and a Novel Strategy in Valvular Heart Disease.Int J Mol Sci. 2025 Feb 18;26(4):1743. doi: 10.3390/ijms26041743. Int J Mol Sci. 2025. PMID: 40004206 Free PMC article. Review.
-
VCAM-1 Targeted Lipopolyplexes as Vehicles for Efficient Delivery of shRNA-Runx2 to Osteoblast-Differentiated Valvular Interstitial Cells; Implications in Calcific Valve Disease Treatment.Int J Mol Sci. 2022 Mar 30;23(7):3824. doi: 10.3390/ijms23073824. Int J Mol Sci. 2022. PMID: 35409184 Free PMC article.
-
Aortic valve disease in diabetes: Molecular mechanisms and novel therapies.J Cell Mol Med. 2021 Oct;25(20):9483-9495. doi: 10.1111/jcmm.16937. Epub 2021 Sep 24. J Cell Mol Med. 2021. PMID: 34561944 Free PMC article. Review.
-
Smart Supra- and Macro-Molecular Tools for Biomedical Applications.Materials (Basel). 2020 Jul 27;13(15):3343. doi: 10.3390/ma13153343. Materials (Basel). 2020. PMID: 32727155 Free PMC article. Review.
References
-
- Yutzey K.E., Demer L.L., Body S.C., Huggins G.S., Towler D.A., Giachelli C.M., Hofmann-Bowman M.A., Mortlock D.P., Rogers M.B., Sadeghi M.M., et al. Calcific aortic valve disease: A consensus summary from the Alliance of Investigators on Calcific Aortic Valve Disease. Arterioscler. Thromb. Vasc. Biol. 2014;34:2387–2393. doi: 10.1161/ATVBAHA.114.302523. - DOI - PMC - PubMed
-
- Virtanen M.P.O., Airaksinen J., Niemela M., Laakso T., Husso A., Jalava M.P., Tauriainen T., Maaranen P., Kinnunen E.M., Dahlbacka S., et al. Comparison of Survival of Transfemoral Transcatheter Aortic Valve Implantation Versus Surgical Aortic Valve Replacement for Aortic Stenosis in Low-Risk Patients Without Coronary Artery Disease. Am. J. Cardiol. 2020;125:589–596. doi: 10.1016/j.amjcard.2019.11.002. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

