Elusive Roles of the Different Ceramidases in Human Health, Pathophysiology, and Tissue Regeneration
- PMID: 32498325
- PMCID: PMC7349419
- DOI: 10.3390/cells9061379
Elusive Roles of the Different Ceramidases in Human Health, Pathophysiology, and Tissue Regeneration
Abstract
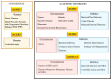
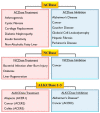
Ceramide and sphingosine are important interconvertible sphingolipid metabolites which govern various signaling pathways related to different aspects of cell survival and senescence. The conversion of ceramide into sphingosine is mediated by ceramidases. Altogether, five human ceramidases-named acid ceramidase, neutral ceramidase, alkaline ceramidase 1, alkaline ceramidase 2, and alkaline ceramidase 3-have been identified as having maximal activities in acidic, neutral, and alkaline environments, respectively. All five ceramidases have received increased attention for their implications in various diseases, including cancer, Alzheimer's disease, and Farber disease. Furthermore, the potential anti-inflammatory and anti-apoptotic effects of ceramidases in host cells exposed to pathogenic bacteria and viruses have also been demonstrated. While ceramidases have been a subject of study in recent decades, our knowledge of their pathophysiology remains limited. Thus, this review provides a critical evaluation and interpretive analysis of existing literature on the role of acid, neutral, and alkaline ceramidases in relation to human health and various diseases, including cancer, neurodegenerative diseases, and infectious diseases. In addition, the essential impact of ceramidases on tissue regeneration, as well as their usefulness in enzyme replacement therapy, is also discussed.
Keywords: ceramidases; ceramides; infectious diseases; inflammation; neurodegenerative diseases.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

