NELL2-mediated lumicrine signaling through OVCH2 is required for male fertility
- PMID: 32499443
- PMCID: PMC7396227
- DOI: 10.1126/science.aay5134
NELL2-mediated lumicrine signaling through OVCH2 is required for male fertility
Abstract
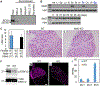
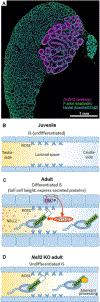
The lumicrine system is a postulated signaling system in which testis-derived (upstream) secreted factors enter the male reproductive tract to regulate epididymal (downstream) pathways required for sperm maturation. Until now, no lumicrine factors have been identified. We demonstrate that a testicular germ-cell-secreted epidermal growth factor-like protein, neural epidermal growth factor-like-like 2 (NELL2), specifically binds to an orphan receptor tyrosine kinase, c-ros oncogene 1 (ROS1), and mediates the differentiation of the initial segment (IS) of the caput epididymis. Male mice in which Nell2 had been knocked out were infertile. The IS-specific secreted proteases, ovochymase 2 (OVCH2) and A disintegrin and metallopeptidase 28 (ADAM28), were expressed upon IS maturation, and OVCH2 was required for processing of the sperm surface protein ADAM3, which is required for sperm fertilizing ability. This work identifies a lumicrine system essential for testis-epididymis-spermatozoa (NELL2-ROS1-OVCH2-ADAM3) signaling and male fertility.
Copyright © 2020 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
Conflict of interest statement
Figures


Comment in
-
Testicular-borne factors affect sperm fertility.Science. 2020 Jun 5;368(6495):1053-1054. doi: 10.1126/science.abc2732. Science. 2020. PMID: 32499425 Free PMC article. No abstract available.
-
Uro-Science.J Urol. 2021 Jan;205(1):307-308. doi: 10.1097/JU.0000000000001447. Epub 2020 Nov 9. J Urol. 2021. PMID: 33164628 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

