cis-4-[18F]fluoro-L-proline Molecular Imaging Experimental Liver Fibrosis
- PMID: 32500081
- PMCID: PMC7243806
- DOI: 10.3389/fmolb.2020.00090
cis-4-[18F]fluoro-L-proline Molecular Imaging Experimental Liver Fibrosis
Abstract
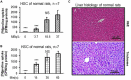
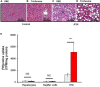
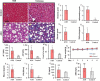
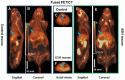
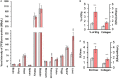
Introduction: Early-stage liver fibrosis is potentially reversible, but difficult to diagnose. Clinical management would be enhanced by the development of a non-invasive imaging technique able to identify hepatic injury early, before end-stage fibrosis ensues. The analog of the amino acid proline, cis-4-[18F]fluoro-L-proline ([18F]fluoro-proline), which targets collagenogenesis in hepatic stellate cells (HSC), was used to detect fibrosis. Methods: Acute steatohepatitis was induced in experimental animals by liquid ethanol diet for 8 weeks, intra-gastric binge feedings every 10th day along with lipopolysaccharide (LPS) injection. The control animals received control diet for 8 weeks and an equivalent volume of saline on the same schedule as the acute steatohepatitis model. First, in vitro cellular experiments were carried out to assess [3H]proline uptake by HSC, hepatocytes and Kupffer cells derived from rats with acute steatohepatitis (n = 14) and controls (n = 14). Next, ex vivo liver experiments were done to investigate unlabeled proline-mediated collagen synthesis and its associated proline transporter expression in acute steatohepatitis (n = 5) and controls (n = 5). Last, in vivo dynamic and static [18F]fluoro-proline micro-PET/CT imaging was performed in animal models of acute steatohepatitis (n = 7) and control (n = 7) mice. Results: [3H]proline uptake was 5-fold higher in the HSCs of steatohepatitis rats than controls after incubation of up to 60 min. There was an excellent correlation between [3H]proline uptake and liver collagen expression (r-value > 0.90, p < 0.05). Subsequent liver tissue studies demonstrated 2-3-fold higher proline transporter expression in acute steatohepatitis animals than in controls, and proline-related collagen synthesis was blocked by this transporter inhibitor. In vivo micro-PET/CT studies with [18F]fluoro-proline showed 2-3-fold higher uptake in the livers of acute steatohepatitis mice than in controls. There was an excellent correlation between [18F]fluoro-proline uptake and liver collagen expression in the livers of acute steatohepatitis mice (r-value = 0.97, p < 0.001). Conclusion: [18F]fluoro-proline localizes in the liver and correlates with collagenogenesis in acute steatohepatitis with a signal intensity that is sufficiently high to allow imaging with micro-PET/CT. Thus, [18F]fluoro-proline could serve as a PET imaging biomarker for detecting early-stage liver fibrosis.
Keywords: PET/CT; cis-4-[18F]fluoro-L-proline; early stage alcoholic liver fibrosis; hepatic stellate cells; molecular imaging; steatohepatitis.
Copyright © 2020 Cao, Lu, Azad, Pomper, Smith, He, Pi, Ren, Ying, Sichani, Morris and Dilsizian.
Figures



References
-
- Affò S. I., Morales-Ibanez O. I., Rodrigo-Torres D. I., Altamirano J. I., Blaya D. I., Dapito D. H. I. I., et al. (2014). CCL20 mediates lipopolysaccharide induced liver injury and is a potential driver of inflammation and fibrosis in alcoholic hepatitis. Gut 63 1782–1792. 10.1136/gutjnl-2013-306098 - DOI - PMC - PubMed
-
- Batey R., Cao Q., Madsen G., Pang G., Russell A., Clancy R. (1998). Decreased tumor necrosis factor-alpha and interleukin-1alpha production from intrahepatic mononuclear cells in chronic ethanol consumption and upregulation by endotoxin. Alcohol. Clin. Exp. Res. 22 150–156. 10.1111/j.1530-0277.1998.tb03631.x - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

