Intracellular Ca2+ Signalling in the Pathogenesis of Acute Pancreatitis: Recent Advances and Translational Perspectives
- PMID: 32503336
- PMCID: PMC7312053
- DOI: 10.3390/ijms21114005
Intracellular Ca2+ Signalling in the Pathogenesis of Acute Pancreatitis: Recent Advances and Translational Perspectives
Abstract
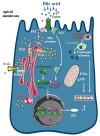
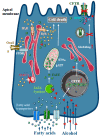
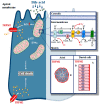
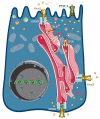
Intracellular Ca2+ signalling is a major signal transductional pathway in non-excitable cells, responsible for the regulation of a variety of physiological functions. In the secretory epithelial cells of the exocrine pancreas, such as acinar and ductal cells, intracellular Ca2+ elevation regulates digestive enzyme secretion in acini or fluid and ion secretion in ductal cells. Although Ca2+ is a uniquely versatile orchestrator of epithelial physiology, unregulated global elevation of the intracellular Ca2+ concentration is an early trigger for the development of acute pancreatitis (AP). Regardless of the aetiology, different forms of AP all exhibit sustained intracellular Ca2+ elevation as a common hallmark. The release of endoplasmic reticulum (ER) Ca2+ stores by toxins (such as bile acids or fatty acid ethyl esters (FAEEs)) or increased intrapancreatic pressure activates the influx of extracellular Ca2+ via the Orai1 Ca2+ channel, a process known as store-operated Ca2+ entry (SOCE). Intracellular Ca2+ overload can lead to premature activation of trypsinogen in pancreatic acinar cells and impaired fluid and HCO3- secretion in ductal cells. Increased and unbalanced reactive oxygen species (ROS) production caused by sustained Ca2+ elevation further contributes to cell dysfunction, leading to mitochondrial damage and cell death. Translational studies of AP identified several potential target molecules that can be modified to prevent intracellular Ca2+ overload. One of the most promising drugs, a selective inhibitor of the Orai1 channel that has been shown to inhibit extracellular Ca2+ influx and protect cells from injury, is currently being tested in clinical trials. In this review, we will summarise the recent advances in the field, with a special focus on the translational aspects of the basic findings.
Keywords: Ca2+ signalling; acinar cell necrosis; acute pancreatitis; bile acid; epithelial ion transport.
Conflict of interest statement
The authors have no conflict of interest to declare.
Figures

References
-
- Peery A.F., Dellon E.S., Lund J., Crockett S.D., McGowan C.E., Bulsiewicz W.J., Gangarosa L.M., Thiny M.T., Stizenberg K., Morgan D.R., et al. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology. 2012;143:1179–1187.e3. doi: 10.1053/j.gastro.2012.08.002. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- GINOP-2.3.2-15-2016-00048 to JM, PD116553 to PP/Hungarian National Research, Development and Innovation Office
- EFOP 3.6.2-16-2017-00006 to JM/Ministry of Human Capacities
- BO/00569/17 to PP, LP2017-18/2017 to JM/Hungarian Academy of Sciences
- 20391-3/2018/FEKUSTRAT to JM/National Excellence Programme
- No. 739593/EU's Horizon 2020 research and innovation program
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

