A new generation of recombinant polypeptides combines multiple protein domains for effective antimicrobial activity
- PMID: 32503648
- PMCID: PMC7275485
- DOI: 10.1186/s12934-020-01380-7
A new generation of recombinant polypeptides combines multiple protein domains for effective antimicrobial activity
Abstract
Background: Although most of antimicrobial peptides (AMPs), being relatively short, are produced by chemical synthesis, several AMPs have been produced using recombinant technology. However, AMPs could be cytotoxic to the producer cell, and if small they can be easily degraded. The objective of this study was to produce a multidomain antimicrobial protein based on recombinant protein nanoclusters to increase the yield, stability and effectivity.
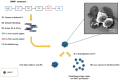
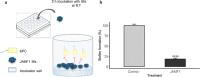
Results: A single antimicrobial polypeptide JAMF1 that combines three functional domains based on human α-defensin-5, human XII-A secreted phospholipase A2 (sPLA2), and a gelsolin-based bacterial-binding domain along with two aggregation-seeding domains based on leucine zippers was successfully produced with no toxic effects for the producer cell and mainly in a nanocluster structure. Both, the nanocluster and solubilized format of the protein showed a clear antimicrobial effect against a broad spectrum of Gram-negative and Gram-positive bacteria, including multi-resistant strains, with an optimal concentration between 1 and 10 µM.
Conclusions: Our findings demonstrated that multidomain antimicrobial proteins forming nanoclusters can be efficiently produced in recombinant bacteria, being a novel and valuable strategy to create a versatile, highly stable and easily editable multidomain constructs with a broad-spectrum antimicrobial activity in both soluble and nanostructured format.
Keywords: Antimicrobial peptides; Antimicrobial resistance; Inclusion bodies; Multidomain protein; Protein nanoclusters; Recombinant production; Solubilization.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
Sequence edition of single domains modulates the final immune and antimicrobial potential of a new generation of multidomain recombinant proteins.Sci Rep. 2021 Dec 10;11(1):23798. doi: 10.1038/s41598-021-03220-z. Sci Rep. 2021. PMID: 34893661 Free PMC article.
-
Beta-defensin derived cationic antimicrobial peptides with potent killing activity against gram negative and gram positive bacteria.BMC Microbiol. 2018 Jun 5;18(1):54. doi: 10.1186/s12866-018-1190-z. BMC Microbiol. 2018. PMID: 29871599 Free PMC article.
-
Comparative Evaluation of the Antimicrobial Activity of Different Antimicrobial Peptides against a Range of Pathogenic Bacteria.PLoS One. 2015 Dec 11;10(12):e0144611. doi: 10.1371/journal.pone.0144611. eCollection 2015. PLoS One. 2015. PMID: 26656394 Free PMC article.
-
An intimate link between antimicrobial peptide sequence diversity and binding to essential components of bacterial membranes.Biochim Biophys Acta. 2016 May;1858(5):958-70. doi: 10.1016/j.bbamem.2015.10.011. Epub 2015 Oct 21. Biochim Biophys Acta. 2016. PMID: 26498397 Review.
-
Structural design and antimicrobial properties of polypeptides and saccharide-polypeptide conjugates.J Mater Chem B. 2020 Oct 21;8(40):9173-9196. doi: 10.1039/d0tb01916j. J Mater Chem B. 2020. PMID: 32954393 Review.
Cited by
-
Antimicrobial Peptides: Mechanism, Expressions, and Optimization Strategies.Probiotics Antimicrob Proteins. 2025 Apr;17(2):857-872. doi: 10.1007/s12602-024-10391-4. Epub 2024 Nov 11. Probiotics Antimicrob Proteins. 2025. PMID: 39528853 Review.
-
Immunomodulation Evidence of Nanostructured Recombinant Proteins in Salmonid Cells.Animals (Basel). 2024 Mar 9;14(6):844. doi: 10.3390/ani14060844. Animals (Basel). 2024. PMID: 38539943 Free PMC article.
-
Exploring the use of leucine zippers for the generation of a new class of inclusion bodies for pharma and biotechnological applications.Microb Cell Fact. 2020 Sep 4;19(1):175. doi: 10.1186/s12934-020-01425-x. Microb Cell Fact. 2020. PMID: 32887587 Free PMC article.
-
Time to consider ruling out inclusion bodies denaturing protocols for spontaneous solubilization of biologically active proteins.Sci Rep. 2024 Oct 30;14(1):26061. doi: 10.1038/s41598-024-77899-1. Sci Rep. 2024. PMID: 39472505 Free PMC article.
-
The dual interaction of antimicrobial peptides on bacteria and cancer cells; mechanism of action and therapeutic strategies of nanostructures.Microb Cell Fact. 2022 Jun 18;21(1):118. doi: 10.1186/s12934-022-01848-8. Microb Cell Fact. 2022. PMID: 35717207 Free PMC article. Review.
References
-
- Haney EF, Mansour SC, Hancock REW. Antimicrobial peptides: an introduction. In: Methods in molecular biology. 2017. p. 3–22. http://www.ncbi.nlm.nih.gov/pubmed/28013493. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

