Fabrication, maturation, and implantation of composite tissue-engineered total discs formed from native and mesenchymal stem cell combinations
- PMID: 32505801
- PMCID: PMC8633846
- DOI: 10.1016/j.actbio.2020.05.039
Fabrication, maturation, and implantation of composite tissue-engineered total discs formed from native and mesenchymal stem cell combinations
Abstract
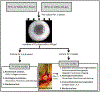
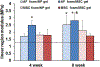
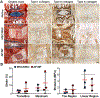
Low back pain arising from disc degeneration is one of the most common causes of limited function in adults. A number of tissue engineering strategies have been used to develop composite tissue engineered total disc replacements to restore native tissue structure and function. In this study we fabricated a composite engineered disc based on the combination of a porous polycaprolactone (PCL) foam annulus fibrosus (AF) and a hyaluronic acid (HA) hydrogel nucleus pulposus (NP). To evaluate whether native tissue cells or mesenchymal stem cells (MSCs) would perform better, constructs were seeded with native AF/NP cells or with MSCs in the foam and/or gel region. Maturation of these composite engineered discs was evaluated for 9 weeks in vitro culture by biochemical content, histological analysis and mechanical properties. To evaluate the performance of these constructs in the in vivo space, engineered discs were implanted into the caudal spines of athymic rats for 5 weeks. Our findings show that engineered discs comprised of AF/NP cells and MSCs performed similarly and maintained their structure after 5 weeks in vivo. However, for both cell types, loss of proteoglycan was evident in the NP region. These data support the continued development of the more clinically relevant MSCs population for disc replacement applications. STATEMENT OF SIGNIFICANCE: A number of tissue engineering strategies have emerged that are focused on the creation of a composite disc replacement. We fabricated a composite engineered disc based on the combination of a porous foam AF and a HA gel NP. We used these constructs to determine whether the combination of AF/NP cells or MSCs would mature to a greater extent in vitro and which cell type would best retain their phenotype after implantation. Engineered discs comprised of AF/NP cells and MSCs performed similarly, maintaining their structure after 5 weeks in vivo. These data support the successful fabrication and in vivo function of an engineered disc composed of a PCL foam AF and a hydrogel NP using either disc cells or MSCs.
Keywords: Disc cells; Intervertebral disc degeneration; Mesenchymal stem cells; Tissue engineering; Total disc replacement.
Copyright © 2020. Published by Elsevier Ltd.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





References
-
- Kauppila LI, Atherosclerosis and disc degeneration/low-back pain- a systematic review, Eur. J. Vasc. Endovasc. Surg 37 (2009) 661–670. - PubMed
-
- Guiot BH, Fessler RG, Molecular biology of degenerative disc disease, Neurosurgery 47 (2000) 1034–1040. - PubMed
-
- Mizuno H, Roy AK, Vacanti CA, Kojima K, Ueda M, Bonassar LJ, Tissue-engineered composites of anulus fibrosus and nucleus pulposus for inter-vertebral disc replacement, Spine 29 (2004) 1290–1298 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

