Hemangioblastoma and von Hippel-Lindau disease: genetic background, spectrum of disease, and neurosurgical treatment
- PMID: 32507909
- PMCID: PMC7575510
- DOI: 10.1007/s00381-020-04712-5
Hemangioblastoma and von Hippel-Lindau disease: genetic background, spectrum of disease, and neurosurgical treatment
Abstract
Introduction: Hemangioblastomas are rare, histologically benign, highly vascularized tumors of the brain, the spinal cord, and the retina, occurring sporadically or associated with the autosomal dominant inherited von Hippel-Lindau (VHL) disease. Children or adults with VHL disease have one of > 300 known germline mutations of the VHL gene located on chromosome 3. They are prone to develop hemangioblastomas, extremely rarely starting at age 6, rarely at age 12-18, and, typically and almost all, as adults. There is a plethora of VHL-associated tumors and cysts, mainly in the kidney, pancreas, adrenals, reproductive organs, and central nervous system. Due to a lack of causal treatment, alleviation of symptoms and prevention of permanent neurological deficits as well as malignant transformation are the main task. Paucity of data and the nonlinear course of tumor progression make management of pediatric VHL patients with hemangioblastomas challenging.
Methods: The Freiburg surveillance protocol was developed by combining data from the literature and our experience of examinations of > 300 VHL patients per year at our university VHL center.
Results: Key recommendations are to start screening of patients at risk by funduscopy with dilated pupils for retinal tumors with admission to school and with MRI of the brain and spinal cord at age 14, then continue biannually until age 18, with emergency MRI in case of neurological symptoms. Indication for surgery remains personalized and should be approved by an experienced VHL board, but we regard neurological symptoms, rapid tumor growth, or critically large tumor/cyst sizes as the key indications to remove hemangioblastomas. Since repeated surgery on hemangioblastomas in VHL patients is not rare, modern neurosurgical techniques should encompass microsurgery, neuronavigation, intraoperative neuromonitoring, fluorescein dye-based intraoperative angiography, intraoperative ultrasound, and minimally invasive approaches, preceded in selected cases by endovascular embolization. Highly specialized neurosurgeons are able to achieve a very low risk of permanent morbidity for the removal of hemangioblastomas from the cerebellum and spinal cord. Small retinal tumors of the peripheral retina can be treated by laser coagulation, larger tumors by cryocoagulation or brachytherapy.
Conclusion: We consider management at experienced VHL centers mandatory and careful surveillance and monitoring of asymptomatic lesions are required to prevent unnecessary operations and minimize morbidity.
Keywords: Adolescence; Childhood; Hemangioblastoma; Screening; Surgery; VHL; von Hippel-Lindau.
Conflict of interest statement
No conflict of interest was reported by the authors.
Figures


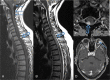



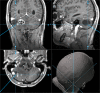
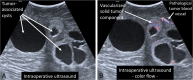

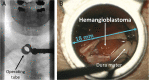

References
-
- Lindau A (1926) Studien über Kleinhirnzysten. Bau, Pathogenese und Beziehungen zur Angiomatosis retinae. Acta Pathol Microbiol Scand Supplement II:1–129
-
- Neumann HPH. Arvid Lindau zum 100. Geburtstag Pathologe. 1993;14:178–180. - PubMed
-
- Plate KH, Aldape KD, Vortmeyer AO, et al. Hemangioblastoma. WHO classification of tumours of the central nervous system. Lyon: IARC Press; 2016.
-
- Vortmeyer AO, Frank S, Jeong S-Y, Yuan K, Ikejiri B, Lee YS, Bhowmick D, Lonser RR, Smith R, Rodgers G, Oldfield EH, Zhuang Z. Developmental arrest of angioblastic lineage initiates tumorigenesis in von Hippel-Lindau disease. Cancer Res. 2003;63:7051–7055. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

