β-Arrestin 1 in Thyrotropin Receptor Signaling in Bone: Studies in Osteoblast-Like Cells
- PMID: 32508750
- PMCID: PMC7251030
- DOI: 10.3389/fendo.2020.00312
β-Arrestin 1 in Thyrotropin Receptor Signaling in Bone: Studies in Osteoblast-Like Cells
Abstract
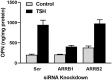
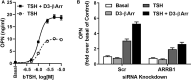
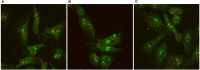
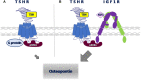
A direct action of thyrotropin (TSH, thyroid-stimulating hormone) on bone precursors in humans is controversial. Studies in rodent models have provided conflicting findings. We used cells derived from a moderately differentiated osteosarcoma stably overexpressing human TSH receptors (TSHRs) as a model of osteoblast precursors (U2OS-TSHR cells) to investigate TSHR-mediated effects in bone differentiation in human cells. We review our findings that (1) TSHR couples to several different G proteins to induce upregulation of genes associated with osteoblast activity-interleukin 11 (IL-11), osteopontin (OPN), and alkaline phosphatase (ALPL) and that the kinetics of the induction and the G protein-mediated signaling pathways involved were different for these genes; (2) TSH can stimulate β-arrestin-mediated signal transduction and that β-arrestin 1 in part mediates TSH-induced pre-osteoblast differentiation; and (3) TSHR/insulin-like growth factor 1 (IGF1) receptor (IGF1R) synergistically increased OPN secretion by TSH and IGF1 and that this crosstalk was mediated by physical association of these receptors in a signaling complex that uses β-arrestin 1 as a scaffold. These findings were complemented using a novel β-arrestin 1-biased agonist of TSHR. We conclude that TSHR can signal via several transduction pathways leading to differentiation of this model system of human pre-osteoblast cells and, therefore, that TSH can directly regulate these bone cells.
Keywords: IGF1 receptor; TSH receptor; crosstalk; osteoblast-like cells; positive allosteric modulator; β-arrestin 1.
Copyright © 2020 Boutin, Gershengorn and Neumann.
Figures

Similar articles
-
β-Arrestin-1 mediates thyrotropin-enhanced osteoblast differentiation.FASEB J. 2014 Aug;28(8):3446-55. doi: 10.1096/fj.14-251124. Epub 2014 Apr 10. FASEB J. 2014. PMID: 24723693 Free PMC article.
-
Discovery of a Positive Allosteric Modulator of the Thyrotropin Receptor: Potentiation of Thyrotropin-Mediated Preosteoblast Differentiation In Vitro.J Pharmacol Exp Ther. 2018 Jan;364(1):38-45. doi: 10.1124/jpet.117.244095. Epub 2017 Oct 31. J Pharmacol Exp Ther. 2018. PMID: 29089368 Free PMC article.
-
Arrestin-β-1 Physically Scaffolds TSH and IGF1 Receptors to Enable Crosstalk.Endocrinology. 2019 Jun 1;160(6):1468-1479. doi: 10.1210/en.2019-00055. Endocrinology. 2019. PMID: 31127272 Free PMC article.
-
The Molecular Function and Clinical Role of Thyroid Stimulating Hormone Receptor in Cancer Cells.Cells. 2020 Jul 20;9(7):1730. doi: 10.3390/cells9071730. Cells. 2020. PMID: 32698392 Free PMC article. Review.
-
Thyrotropin receptor antibodies: new insights into their actions and clinical relevance.Best Pract Res Clin Endocrinol Metab. 2005 Mar;19(1):33-52. doi: 10.1016/j.beem.2004.11.005. Best Pract Res Clin Endocrinol Metab. 2005. PMID: 15826921 Review.
Cited by
-
Roles of IL-11 in the regulation of bone metabolism.Front Endocrinol (Lausanne). 2024 Jan 30;14:1290130. doi: 10.3389/fendo.2023.1290130. eCollection 2023. Front Endocrinol (Lausanne). 2024. PMID: 38352248 Free PMC article. Review.
-
Graves' Autoantibodies Exhibit Different Stimulating Activities in Cultures of Thyrocytes and Orbital Fibroblasts Not Reflected by Clinical Assays.Thyroid. 2022 Jan;32(1):90-96. doi: 10.1089/thy.2021.0326. Epub 2021 Nov 29. Thyroid. 2022. PMID: 34714162 Free PMC article.
-
TSH enhances neurite outgrowth.Front Endocrinol (Lausanne). 2024 Oct 17;15:1463964. doi: 10.3389/fendo.2024.1463964. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39483982 Free PMC article.
-
Thyrotropin, Hyperthyroidism, and Bone Mass.J Clin Endocrinol Metab. 2021 Nov 19;106(12):e4809-e4821. doi: 10.1210/clinem/dgab548. J Clin Endocrinol Metab. 2021. PMID: 34318885 Free PMC article. Review.
-
Bone circuitry and interorgan skeletal crosstalk.Elife. 2023 Jan 19;12:e83142. doi: 10.7554/eLife.83142. Elife. 2023. PMID: 36656634 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

