Progress in the Enzymology of the Mitochondrial Diseases of Lipoic Acid Requiring Enzymes
- PMID: 32508887
- PMCID: PMC7253636
- DOI: 10.3389/fgene.2020.00510
Progress in the Enzymology of the Mitochondrial Diseases of Lipoic Acid Requiring Enzymes
Abstract
Three human mitochondrial diseases that directly affect lipoic acid metabolism result from heterozygous missense and nonsense mutations in the LIAS, LIPT1, and LIPT2 genes. However, the functions of the proteins encoded by these genes in lipoic acid metabolism remained uncertain due to a lack of biochemical analysis at the enzyme level. An exception was the LIPT1 protein for which a perplexing property had been reported, a ligase lacking the ability to activate its substrate. This led to several models, some contradictory, to accommodate the role of LIPT1 protein activity in explaining the phenotypes of the afflicted neonatal patients. Recent evidence indicates that this LIPT1 protein activity is a misleading evolutionary artifact and that the physiological role of LIPT1 is in transfer of lipoic acid moieties from one protein to another. This and other new biochemical data now define a straightforward pathway that fully explains each of the human disorders specific to the assembly of lipoic acid on its cognate enzyme proteins.
Keywords: LIAS; LIPT1; LIPT2; glycine cleavage system; lipoate assembly; lipoic acid; pyruvate dehydrogenase; α-ketoglutarate dehydrogenase.
Copyright © 2020 Cronan.
Figures
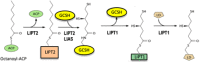
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

