Kisspeptin and neurokinin B interactions in modulating gonadotropin secretion in women with polycystic ovary syndrome
- PMID: 32510130
- PMCID: PMC7316500
- DOI: 10.1093/humrep/deaa104
Kisspeptin and neurokinin B interactions in modulating gonadotropin secretion in women with polycystic ovary syndrome
Abstract
Study question: What is the role of the hypothalamic neuropeptide neurokinin B (NKB) and its interaction with kisspeptin on GnRH/LH secretion in women with polycystic ovary syndrome (PCOS)?
Summary answer: Administration of neurokinin 3 receptor antagonist (NK3Ra) for 7 days reduced LH and FSH secretion and LH pulse frequency in women with PCOS, whilst the stimulatory LH response to kisspeptin-10 was maintained.
What is known already: PCOS is characterized by abnormal GnRH/LH secretion. NKB and kisspeptin are master regulators of GnRH/LH secretion, but their role in PCOS is unclear.
Study design, size, duration: The NK3Ra MLE4901, 40 mg orally twice a day, was administered to women with PCOS for 7 days (n = 8) (vs no treatment, n = 7). On the last day of NK3Ra administration or the equivalent day in those not treated, women were randomized to 7-h kisspeptin-10 (4 µg/kg/h i.v.) or vehicle infusion. This was repeated with the alternate infusion in a subsequent cycle.
Participants/materials, setting, methods: Subjects were women with PCOS, studied in a Clinical Research Facility. Reproductive hormones were measured before and after NK3Ra administration. On the last day of NK3Ra administration (or the equivalent cycle day in untreated women), all women attended for an 8-h frequent blood sampling to allow analysis of the pulsatile LH secretion.
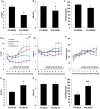
Main results and the role of chance: NK3Ra reduced LH secretion (4.0 ± 0.4 vs 6.5 ± 0.8 IU/l, P < 0.05) and pulse frequency (0.5 ± 0.1 vs 0.8 ± 0.1 pulses/h, P < 0.05); FSH secretion was also reduced (2.0 ± 0.3 vs 2.5 ± 0.4 IU/l, P < 0.05). Without NK3Ra pre-treatment, kisspeptin-10 increased LH secretion (5.2 ± 0.5 to 7.8 ± 1.0 IU/L, P < 0.05), with a positive relationship to oestradiol concentrations (r2 = 0.59, P < 0.05). After NK3Ra administration, the LH response to kisspeptin-10 was preserved (vehicle 3.5 ± 0.3 vs 9.0 ± 2.2 IU/l with kisspeptin-10, P < 0.05), but the positive correlation with oestradiol concentrations was abolished (r2 = 0.07, ns. after NK3Ra). FSH secretion was increased by kisspeptin-10 after NK3Ra treatment, but not without NK3Ra treatment.
Limitations, reasons for caution: The study did not explore the dose relationship of the effect of NK3R antagonism. The impact of obesity or other aspects of the variability of the PCOS phenotype was not studied due to the small number of subjects.
Wider implications of the findings: These data demonstrate the interactive regulation of GnRH/LH secretion by NKB and kisspeptin in PCOS, and that the NKB system mediates aspects of oestrogenic feedback.
Study funding/competing interest(s): Wellcome Trust through Scottish Translational Medicine and Therapeutics Initiative (102419/Z/13/A) and MRC grants (G0701682 to R.P.M. and R.A.A.) and MR/N022556/1 to the MRC Centre for Reproductive Health. This work was performed within the Edinburgh Clinical Research Facility. J.T.G. has undertaken consultancy work for AstraZeneca and Takeda Pharmaceuticals and is an employee of Boehringer Ingelheim. R.P.M. has consulted for Ogeda and was CEO of Peptocrine. R.A.A. has undertaken consultancy work for Merck, Ferring, NeRRe Therapeutics and Sojournix Inc. J.D.V. and K.S. have nothing to disclose.
Trial registration number: N/A.
Keywords: neurokinin B / kisspeptin / LH pulsatility / PCOS / GnRH.
© The Author(s) 2020. Published by Oxford University Press on behalf of European Society of Human Reproduction and Embryology.
Figures


References
-
- Abbara A, Jayasena CN, Christopoulos G, Narayanaswamy S, Izzi-Engbeaya C,, Nijher GM, Comninos AN, Peters D, Buckley A, Ratnasabapathy R. et al. Efficacy of Kisspeptin-54 to trigger oocyte maturation in women at high risk of ovarian hyperstimulation syndrome (OHSS) during in vitro fertilization (IVF) therapy. J Clin Endocrinol Metab 2015;100:3322–3331. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

