This is a preprint.
Clinical benefit of remdesivir in rhesus macaques infected with SARS-CoV-2
- PMID: 32511319
- PMCID: PMC7239049
- DOI: 10.1101/2020.04.15.043166
Clinical benefit of remdesivir in rhesus macaques infected with SARS-CoV-2
Update in
-
Clinical benefit of remdesivir in rhesus macaques infected with SARS-CoV-2.Nature. 2020 Sep;585(7824):273-276. doi: 10.1038/s41586-020-2423-5. Epub 2020 Jun 9. Nature. 2020. PMID: 32516797 Free PMC article.
Abstract
Background: Effective therapeutics to treat COVID-19 are urgently needed. Remdesivir is a nucleotide prodrug with in vitro and in vivo efficacy against coronaviruses. Here, we tested the efficacy of remdesivir treatment in a rhesus macaque model of SARS-CoV-2 infection.
Methods: To evaluate the effect of remdesivir treatment on SARS-CoV-2 disease outcome, we used the recently established rhesus macaque model of SARS-CoV-2 infection that results in transient lower respiratory tract disease. Two groups of six rhesus macaques were infected with SARS-CoV-2 and treated with intravenous remdesivir or an equal volume of vehicle solution once daily. Clinical, virological and histological parameters were assessed regularly during the study and at necropsy to determine treatment efficacy.
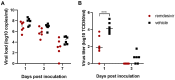
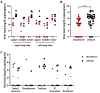
Results: In contrast to vehicle-treated animals, animals treated with remdesivir did not show signs of respiratory disease and had reduced pulmonary infiltrates on radiographs. Virus titers in bronchoalveolar lavages were significantly reduced as early as 12hrs after the first treatment was administered. At necropsy on day 7 after inoculation, lung viral loads of remdesivir-treated animals were significantly lower and there was a clear reduction in damage to the lung tissue.
Conclusions: Therapeutic remdesivir treatment initiated early during infection has a clear clinical benefit in SARS-CoV-2-infected rhesus macaques. These data support early remdesivir treatment initiation in COVID-19 patients to prevent progression to severe pneumonia.
Conflict of interest statement
Conflict of interest The authors affiliated with Gilead Sciences are employees of the company and own company stock. The authors affiliated with NIH have no conflict of interest to report.
Figures




References
-
- Remdesivir. 2020. (Accessed 04/09/2020, at https://clinicaltrials.gov/ct2/results?cond=&term=remdesivir&cntry=&stat...)
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
