Evaluation of ViroTrack Sero Zika IgG/IgM, a New Rapid and Quantitative Zika Serological Diagnostic Assay
- PMID: 32512812
- PMCID: PMC7345115
- DOI: 10.3390/diagnostics10060372
Evaluation of ViroTrack Sero Zika IgG/IgM, a New Rapid and Quantitative Zika Serological Diagnostic Assay
Abstract
Dengue virus (DENV) and Zika virus (ZIKV) belong to the flavivirus genus and are antigenically closely related. They also share the same mosquito vector and can cause similar symptoms upon infection. However, DENV and ZIKV infections lead to different clinical sequelae and treatments; therefore, clinicians need rapid and accurate diagnostics capable of distinguishing between the two diseases.
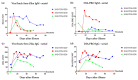
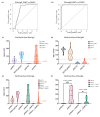
Methods: We employed the immuno-magnetic assay technology on a microfluidic cartridge (ViroTrack Sero Zika IgG/IgM) for diagnosis of ZIKV infection based on the aggregation of magnetic nanoparticles. We carried out three serological studies including samples from the Dominican Republic, USA, and Nicaragua, aimed at detecting ZIKV-specific IgG and IgM using the ViroTrack Sero Zika IgG/IgM test.
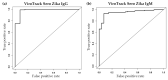
Results: The seroconversion results were comparable with ZIKV IgG and IgM reactivity measured by the commercial ZIKV ELISA kit. The sensitivity and specificity for both ZIKV IgG and IgM tested by the ViroTrack Sero Zika IgG/IgM was approximately 98% and 93%, respectively.
Conclusion: Serological detection of ZIKV infection by the new ViroTrack Sero Zika IgG/IgM test shows promising performance and limited cross-reactivity with DENV.
Keywords: ViroTrack Sero Zika IgG/IgM; Zika virus; dengue virus; diagnosis; immuno-magnetic assay.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Evaluation of a new rapid fluorescence immunoassay for the diagnosis of dengue and Zika virus infection.J Clin Virol. 2019 Mar;112:34-39. doi: 10.1016/j.jcv.2019.01.011. Epub 2019 Jan 29. J Clin Virol. 2019. PMID: 30738366
-
Serological tests reveal significant cross-reactive human antibody responses to Zika and Dengue viruses in the Mexican population.Acta Trop. 2020 Jan;201:105201. doi: 10.1016/j.actatropica.2019.105201. Epub 2019 Sep 25. Acta Trop. 2020. PMID: 31562846
-
Validation of serological and molecular methods for diagnosis of zika virus infections.J Virol Methods. 2019 Jan;263:68-74. doi: 10.1016/j.jviromet.2018.10.011. Epub 2018 Oct 18. J Virol Methods. 2019. PMID: 30342068
-
Does prior dengue virus exposure worsen clinical outcomes of Zika virus infection? A systematic review, pooled analysis and lessons learned.PLoS Negl Trop Dis. 2019 Jan 25;13(1):e0007060. doi: 10.1371/journal.pntd.0007060. eCollection 2019 Jan. PLoS Negl Trop Dis. 2019. PMID: 30682026 Free PMC article.
-
Seroprevalence of Dengue and Zika Virus in Blood Donations: A Systematic Review.Transfus Med Rev. 2019 Jan;33(1):35-42. doi: 10.1016/j.tmrv.2018.10.001. Epub 2018 Oct 31. Transfus Med Rev. 2019. PMID: 30471867
Cited by
-
Evaluation of the Diagnostic Accuracy of a New Biosensors-Based Rapid Diagnostic Test for the Point-Of-Care Diagnosis of Previous and Recent Dengue Infections in Malaysia.Biosensors (Basel). 2021 Apr 22;11(5):129. doi: 10.3390/bios11050129. Biosensors (Basel). 2021. PMID: 33921935 Free PMC article.
-
Serological Evidence of Widespread Zika Transmission across the Philippines.Viruses. 2021 Jul 23;13(8):1441. doi: 10.3390/v13081441. Viruses. 2021. PMID: 34452307 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources

