Multinucleated giant cells within the in vivo implantation bed of a collagen-based biomaterial determine its degradation pattern
- PMID: 32514904
- PMCID: PMC7878236
- DOI: 10.1007/s00784-020-03373-7
Multinucleated giant cells within the in vivo implantation bed of a collagen-based biomaterial determine its degradation pattern
Abstract
Objectives: The aim of the present study was to characterize the cellular reaction to a xenogeneic resorbable collagen membrane of porcine origin using a subcutaneous implantation model in Wistar rats over 30 days.
Materials and methods: Ex vivo, liquid platelet-rich fibrin (PRF), a leukocyte and platelet-rich cell suspension, was used to evaluate the blood cell membrane interaction. The material was implanted subcutaneously in rats. Sham-operated rats without biomaterial displayed physiological wound healing (control group). Histological, immunohistological, and histomorphometric analyses were focused on the inflammatory pattern, vascularization rate, and degradation pattern.
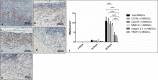
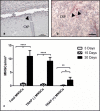
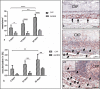
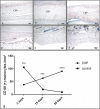
Results: The membrane induced a large number of mononuclear cells over the observation period, including lymphocytes, macrophages, and fibroblasts. After 15 days, multinucleated giant cells (MNGCs) were observed on the biomaterial surface. Their number increased significantly, and they proceeded to the center of the biomaterial on day 30. These cells highly expressed CD-68, calcitonin receptor, and MMP-9, but not TRAP or integrin-ß3. Thus, the membrane lost its integrity and underwent disintegration as a consequence of the induction of MNGCs. The significant increase in MNGC number correlated with a high rate of vascularization, which was significantly higher than the control group. Physiological wound healing in the control group did not induce any MNGCs at any time point. Ex vivo blood cells from liquid-PRF did not penetrate the membrane.
Conclusion: The present study suggests a potential role for MNGCs in biomaterial degradation and questions whether it is beneficial to accept them in clinically approved biomaterials or focus on biomaterials that induce only mononuclear cells. Thus, further studies are necessary to identify the function of biomaterial-induced MNGCs.
Clinical relevance: Understanding the cellular reaction to biomaterials is essential to assess their suitability for specific clinical indications and outline the potential benefit of specific group of biomaterials in the respective clinical indications.
Keywords: Disintegration; Guided bone regeneration (GBR); Guided tissue regeneration (GTR); Inflammatory pattern; Integration.
Conflict of interest statement
The authors declare that they have no conflict of interests.
Figures


Similar articles
-
Modification of collagen-based sponges can induce an upshift of the early inflammatory response and a chronic inflammatory reaction led by M1 macrophages: an in vivo study.Clin Oral Investig. 2020 Oct;24(10):3485-3500. doi: 10.1007/s00784-020-03219-2. Epub 2020 Feb 17. Clin Oral Investig. 2020. PMID: 32065310
-
Sugar-based collagen membrane cross-linking increases barrier capacity of membranes.Clin Oral Investig. 2018 May;22(4):1851-1863. doi: 10.1007/s00784-017-2281-1. Epub 2017 Dec 6. Clin Oral Investig. 2018. PMID: 29209937
-
In vivo Implantation of a Bovine-Derived Collagen Membrane Leads to Changes in the Physiological Cellular Pattern of Wound Healing by the Induction of Multinucleated Giant Cells: An Adverse Reaction?Front Bioeng Biotechnol. 2018 Aug 14;6:104. doi: 10.3389/fbioe.2018.00104. eCollection 2018. Front Bioeng Biotechnol. 2018. PMID: 30155464 Free PMC article.
-
In vivo cellular reactions to different biomaterials-Physiological and pathological aspects and their consequences.Semin Immunol. 2017 Feb;29:49-61. doi: 10.1016/j.smim.2017.06.001. Epub 2017 Jun 21. Semin Immunol. 2017. PMID: 28647227 Review.
-
Giant cells around bone biomaterials: Osteoclasts or multi-nucleated giant cells?Acta Biomater. 2016 Dec;46:15-28. doi: 10.1016/j.actbio.2016.09.029. Epub 2016 Sep 22. Acta Biomater. 2016. PMID: 27667014 Review.
Cited by
-
Effects on Tissue Integration of Collagen Scaffolds Used for Local Delivery of Gentamicin in a Rat Mandible Defect Model.Bioengineering (Basel). 2022 Jun 24;9(7):275. doi: 10.3390/bioengineering9070275. Bioengineering (Basel). 2022. PMID: 35877326 Free PMC article.
-
Interactions Between Immunomodulatory Biomaterials and Immune Microenvironment: Cues for Immunomodulation Strategies in Tissue Repair.Front Bioeng Biotechnol. 2022 May 13;10:820940. doi: 10.3389/fbioe.2022.820940. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35646833 Free PMC article. Review.
-
Mesenchymal Stem Cells Combined with a P(VDF-TrFE)/BaTiO3 Scaffold and Photobiomodulation Therapy Enhance Bone Repair in Rat Calvarial Defects.J Funct Biomater. 2023 Jun 1;14(6):306. doi: 10.3390/jfb14060306. J Funct Biomater. 2023. PMID: 37367270 Free PMC article.
-
In Vivo Biocompatibility Analysis of a Novel Barrier Membrane Based on Bovine Dermis-Derived Collagen for Guided Bone Regeneration (GBR).Membranes (Basel). 2022 Mar 30;12(4):378. doi: 10.3390/membranes12040378. Membranes (Basel). 2022. PMID: 35448348 Free PMC article.
-
The Early Fragmentation of a Bovine Dermis-Derived Collagen Barrier Membrane Contributes to Transmembraneous Vascularization-A Possible Paradigm Shift for Guided Bone Regeneration.Membranes (Basel). 2021 Mar 9;11(3):185. doi: 10.3390/membranes11030185. Membranes (Basel). 2021. PMID: 33803205 Free PMC article.
References
-
- Karring T. The role of bone regeneration in post-surgical periodontal wound healing. Orthod Fr. 1986;57 Pt 2:435–442. - PubMed
-
- Dahlin C, Sennerby L, Lekholm U, Linde A, Nyman S. Generation of new bone around titanium implants using a membrane technique: an experimental study in rabbits. Int J Oral Maxillofac Implants. 1989;4:19–25. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

