Detection of Colorectal Cancer and Advanced Adenoma by Liquid Biopsy (Decalib Study): The ddPCR Challenge
- PMID: 32517177
- PMCID: PMC7352444
- DOI: 10.3390/cancers12061482
Detection of Colorectal Cancer and Advanced Adenoma by Liquid Biopsy (Decalib Study): The ddPCR Challenge
Abstract
Background: In most countries, participation in colorectal cancer (CRC) screening programs with the immunological fecal occult blood test (iFOBT) is low. Mutations of RAS and BRAF occur early in colorectal carcinogenesis and "liquid biopsy" allows detection of mutated circulating tumor DNA (ctDNA). This prospective study aims to evaluate the performance of RAS and BRAF-mutated ctDNA in detecting CRC and advanced adenomas (AA).
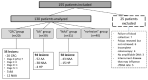
Methods: One hundred and thirty patients who underwent colonoscopy for suspicion of colorectal lesion were included and divided into four groups: 20 CRC, 39 AA, 31 non-advanced adenoma and/or hyperplastic polyp(s) (NAA) and 40 with no lesion. Mutated ctDNA was analyzed by droplet digital PCR.
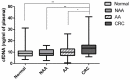
Results: ctDNA was detected in 45.0% of CRC, in 2.6% of AA and none of the NAA and "no-lesion" groups. All patients with stage II to IV mutated CRC had detectable ctDNA (n = 8/8). Among the mutated AA, only one patient had detectable ctDNA (4.3%), maybe due to limited technical sensitivity or to a low rate of ctDNA or even the absence ctDNA in plasma. Specificity and sensitivity of KRAS- and BRAF-mutated ctDNA for the detection of all CRC and AA were 100% and 16.9%, respectively.
Conclusions: ctDNA had high sensitivity in detection of advanced mutated CRC but was unable to sensitively detect AA. ctDNA analysis was easy to perform and readily accepted by the population but requires combination with other circulating biomarkers before replacing iFOBT.
Keywords: circulating tumor DNA; colorectal cancer; droplet digital PCR; fecal occult blood test; liquid biopsy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Multiplex detection of ctDNA mutations in plasma of colorectal cancer patients by PCR/SERS assay.Nanotheranostics. 2020 Aug 25;4(4):224-232. doi: 10.7150/ntno.48905. eCollection 2020. Nanotheranostics. 2020. PMID: 32923312 Free PMC article.
-
Detection of circulating tumor DNA in patients of operative colorectal and gastric cancers.Oncotarget. 2020 Aug 25;11(34):3198-3207. doi: 10.18632/oncotarget.27682. eCollection 2020 Aug 25. Oncotarget. 2020. PMID: 32922660 Free PMC article.
-
Circulating tumor DNA-based early detection of precancerous colorectal lesions using QClamp XNA-mediated real-time PCR.Pharmazie. 2021 Dec 5;76(12):606-610. doi: 10.1691/ph.2021.1780. Pharmazie. 2021. PMID: 34986957
-
[Chemical or immunological tests for the detection of fecal occult blood in colorectal cancer screening?].Gastroenterol Hepatol. 2009 Oct;32(8):565-76. doi: 10.1016/j.gastrohep.2009.01.179. Epub 2009 Jul 3. Gastroenterol Hepatol. 2009. PMID: 19577340 Review. Spanish.
-
[Liquid biopsy in colorectal cancer : An overview of ctDNA analysis in tumour diagnostics].Pathologe. 2019 Dec;40(Suppl 3):244-251. doi: 10.1007/s00292-019-00698-3. Pathologe. 2019. PMID: 31797045 Review. German.
Cited by
-
Decoding the Dynamics of Circulating Tumor DNA in Liquid Biopsies.Cancers (Basel). 2024 Jul 1;16(13):2432. doi: 10.3390/cancers16132432. Cancers (Basel). 2024. PMID: 39001494 Free PMC article. Review.
-
Discovery of extracellular vesicle-delivered miR-185-5p in the plasma of patients as an indicator for advanced adenoma and colorectal cancer.J Transl Med. 2023 Jun 29;21(1):421. doi: 10.1186/s12967-023-04249-6. J Transl Med. 2023. PMID: 37386465 Free PMC article.
-
Research trends and prospects of Circulating tumor DNA associated with colorectal cancer: a bibliometric analysis.Discov Oncol. 2025 Aug 18;16(1):1575. doi: 10.1007/s12672-025-03394-2. Discov Oncol. 2025. PMID: 40824408 Free PMC article.
-
Ultrasensitive plasma-based monitoring of tumor burden using machine-learning-guided signal enrichment.Nat Med. 2024 Jun;30(6):1655-1666. doi: 10.1038/s41591-024-03040-4. Epub 2024 Jun 14. Nat Med. 2024. PMID: 38877116 Free PMC article.
-
Early screening of colorectal cancer using feature engineering with artificial intelligence-enhanced analysis of nanoscale chromatin modifications.Res Sq [Preprint]. 2023 Oct 31:rs.3.rs-3500134. doi: 10.21203/rs.3.rs-3500134/v1. Res Sq. 2023. Update in: Sci Rep. 2024 Apr 2;14(1):7808. doi: 10.1038/s41598-024-58016-8. PMID: 37961494 Free PMC article. Updated. Preprint.
References
-
- Lieberman D.A., Rex D.K., Winawer S.J., Giardiello F.M., Johnson D.A., Levin T.R. Guidelines for Colonoscopy Surveillance After Screening and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2012;143:844–857. doi: 10.1053/j.gastro.2012.06.001. - DOI - PubMed
-
- Denis B., Guittet L. Dépistage du cancer colorectal par test immunologique quantitatif de recherche de sang occulte dans les selles: Une révolution? Hépato-Gastro. Oncol. Dig. 2015;22:119–129. doi: 10.1684/hpg.2014.1113. - DOI
-
- Park D.I., Ryu S., Kim Y.-H., Lee S.-H., Lee C.K., Eun C.S., Han D.S. Comparison of Guaiac-Based and Quantitative Immunochemical Fecal Occult Blood Testing in a Population at Average Risk Undergoing Colorectal Cancer Screening. Am. J. Gastroenterol. 2010;105:2017–2025. doi: 10.1038/ajg.2010.179. - DOI - PubMed
-
- Vitellius C., Laly M., Banaszuk A.-S., Deherce I., Cornet N., Bertrais S., Saulnier P., Caroli-Bosc F.-X. Contribution of the OC Sensor® immunoassay in comparison to the Hemoccult II® guaiac-test in organized colorectal cancer screening. Eur. J. Epidemiol. 2018 doi: 10.1007/s10654-018-0471-z. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

