Phase I study of vemurafenib in children with recurrent or progressive BRAFV600E mutant brain tumors: Pacific Pediatric Neuro-Oncology Consortium study (PNOC-002)
- PMID: 32523649
- PMCID: PMC7260122
- DOI: 10.18632/oncotarget.27600
Phase I study of vemurafenib in children with recurrent or progressive BRAFV600E mutant brain tumors: Pacific Pediatric Neuro-Oncology Consortium study (PNOC-002)
Abstract
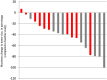
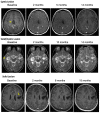
Background: BRAFV600E mutation is present in a subset of pediatric brain tumors. Vemurafenib is an oral, selective ATP-competitive inhibitor of BRAFV600E kinase. The goal of this multi-center study conducted through the Pacific Pediatric Neuro-Oncology Consortium (PNOC) was to determine the recommended phase 2 dose (RP2D) and dose limiting toxicities (DLTs) in children < 18 years with recurrent or progressive BRAFV600E mutant brain tumors. Results: Nineteen eligible patients were enrolled. Eleven patients had received three or more prior therapies. Data reported are from the start of treatment for the first patient (April 30 2014) through August 31 2019. The RP2D was defined as 550 mg/m2 twice daily after DLT criteria adjustment for rash. Related grade ≥ 3 adverse events included secondary keratoacanthoma (n = 1); rash (n =16); and fever (n = 5). Subjects received a median of 23 cycles (range 3-63). Four patients remain on treatment. Centrally reviewed best radiographic responses included 1 complete response, 5 partial responses, and 13 stable disease. The steady-state area under the curve (AUC0-∞median) was 604 mg*h/L (range 329-1052). Methods: Vemurafenib was given starting at 550 mg/m2, twice daily which corresponds to the adult RP2D. Adverse events were graded using the NIH Common Terminology Criteria for Adverse Events (CTCAE) version 4.0. Central imaging review was performed. Pharmacokinetic sampling was performed. Conclusions: Vemurafenib has promising anti-tumor activity in recurrent BRAF V600E-positive brain tumors with manageable toxicity. A phase 2 study is ongoing (NCT01748149).
Keywords: BRAFV600E; clinical trial; pediatric glioma; vemurafenib.
Copyright: © 2020 Nicolaides et al.
Conflict of interest statement
CONFLICTS OF INTEREST There are no conflicts of interest for any authors.
Figures
References
-
- Cammareri P, Rose AM, Vincent DF, Wang J, Nagano A, Libertini S, Ridgway RA, Athineos D, Coates PJ, McHugh A, Pourreyron C, Dayal JH, Larsson J, et al. . Inactivation of TGFbeta receptors in stem cells drives cutaneous squamous cell carcinoma. Nat Commun. 2016; 7:12493. 10.1038/ncomms12493. - DOI - PMC - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

