Allosteric control of hemoglobin S fiber formation by oxygen and its relation to the pathophysiology of sickle cell disease
- PMID: 32527859
- PMCID: PMC7334536
- DOI: 10.1073/pnas.1922004117
Allosteric control of hemoglobin S fiber formation by oxygen and its relation to the pathophysiology of sickle cell disease
Abstract
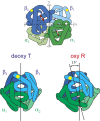
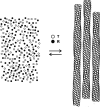
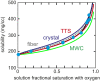
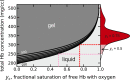
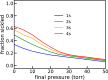
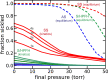
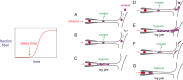
The pathology of sickle cell disease is caused by polymerization of the abnormal hemoglobin S upon deoxygenation in the tissues to form fibers in red cells, causing them to deform and occlude the circulation. Drugs that allosterically shift the quaternary equilibrium from the polymerizing T quaternary structure to the nonpolymerizing R quaternary structure are now being developed. Here we update our understanding on the allosteric control of fiber formation at equilibrium by showing how the simplest extension of the classic quaternary two-state allosteric model of Monod, Wyman, and Changeux to include tertiary conformational changes provides a better quantitative description. We also show that if fiber formation is at equilibrium in vivo, the vast majority of cells in most tissues would contain fibers, indicating that it is unlikely that the disease would be survivable once the nonpolymerizing fetal hemoglobin has been replaced by adult hemoglobin S at about 1 y after birth. Calculations of sickling times, based on a recently discovered universal relation between the delay time prior to fiber formation and supersaturation, show that in vivo fiber formation is very far from equilibrium. Our analysis indicates that patients survive because the delay period allows the majority of cells to escape the small vessels of the tissues before fibers form. The enormous sensitivity of the duration of the delay period to intracellular hemoglobin composition also explains why sickle trait, the heterozygous condition, and the compound heterozygous condition of hemoglobin S with pancellular hereditary persistence of fetal hemoglobin are both relatively benign conditions.
Keywords: polymerization; protein fibers; sickle cell.
Copyright © 2020 the Author(s). Published by PNAS.
Conflict of interest statement
The authors declare no competing interest.
Figures

References
-
- Bunn H. F., Forget B. G., Hemoglobin: Molecular, Genetic and Clinical Aspects, (W. B. Saunders Company, 1986), p. 690.
-
- Pauling L., Itano H. A., Singer S. J., Wells I. C., Sickle cell anemia a molecular disease. Science 110, 543–548 (1949). - PubMed
-
- Eaton W. A., Linus Pauling and sickle cell disease. Biophys. Chem. 100, 109–116 (2003). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

